Transition of MET-NSCLC From an “Untapped” Segment to One With Significant Pipeline Activity
May 08, 2026
Table of Contents
Non-small Cell Lung Cancer (NSCLC) treatment has evolved from a chemotherapy-dominated approach to a biomarker-driven paradigm, enabled by the identification of multiple actionable alterations, including the EGFR signaling pathway, ALK, BRAF, RET, NTRK, HER2, KRAS, ROS1, and MET alterations. This shift has expanded targeted NSCLC treatment options and improved outcomes; however, resistance and suboptimal durability remain persistent challenges. Biomarker testing is crucial for all patients with advanced NSCLC to identify actionable genetic mutations (EGFR, ALK, ROS1, BRAF, MET, RET, NTRK, KRAS) and protein expression (PD-L1). It is recommended to use comprehensive Next-generation Sequencing (NGS) upfront to guide targeted therapy or immunotherapy, improving survival rates.
MET Biomarker Testing Rate
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Evotec, Takeda Deal; AZ COVID Vaccine US Trials; Promega’s Recognition; Roche’s Tecentriq R...
- Lilly’s Morphic Acquisition; IDEAYA’s IDE397 Positive Phase II Trial Result; XPOVIO (seline...
- Imfinzi fails. What’s next in Non-small cell lung cancers Market Scenario?
- Incyte’s ZYNYZ Approved as First-Line Treatment for Advanced Anal Cancer; Amneal Gets FDA Nod for...
- C4X Discovery and AstraZeneca Signs Deal; FDA Rejects Spectrum’s Poziotinib; Orphan Drug Designat...
- Data suggest that over 70% of NSCLC patients are now tested for c-MET alterations in the United States; the rates in Europe are lower than in the United States.
- Reason for Improvement: Growing awareness of MET-targeted therapies, wider adoption of NGS, and better guideline integration.
- Implication: Despite progress, a significant proportion of NSCLC patients still lack testing, highlighting the need for universal, comprehensive genomic profiling to ensure eligible patients receive precision therapy.
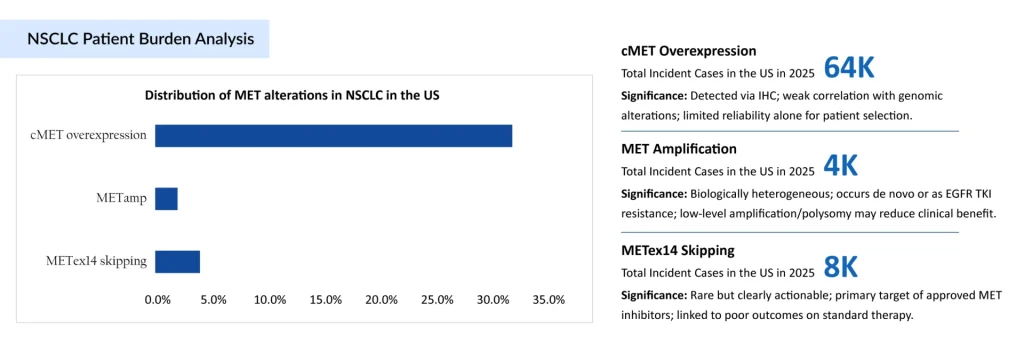
Insights Around NSCLC Patient Burden
MET-NSCLC has transitioned from a neglected niche to one of the most dynamic segments in lung cancer research. MET alterations in NSCLC span overexpression (MET OE), amplification (MET AMP), and MET exon 14 skipping mutations, but clinical relevance varies significantly across these subsets. As per the DelveInsight assessment, the total incident cases of c-MET NSCLC were around 77,000 in 2025 in the United States. These c-MET NSCLC cases are projected to increase during the forecast period (2026 ̶ 2036).
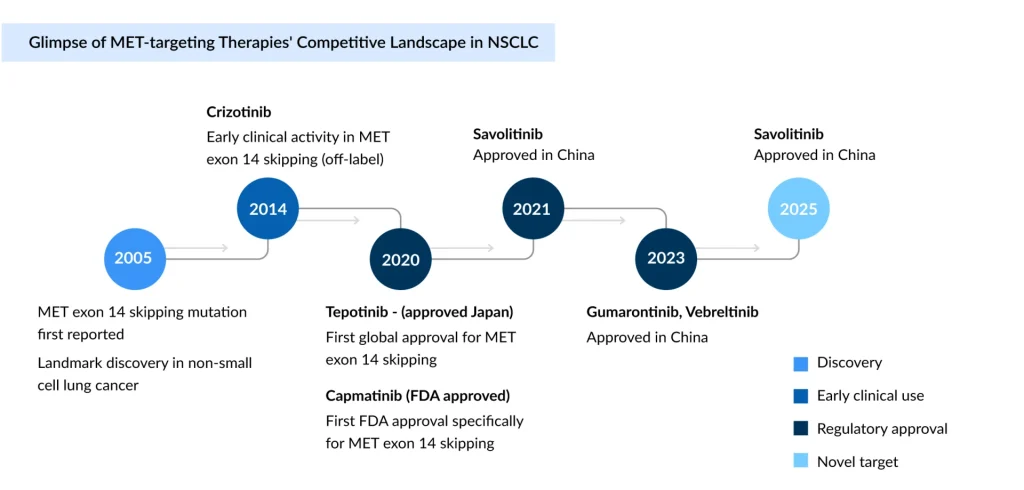
From Untapped Target to Treatment: c‑MET-directed Approved Therapies
Before 2020, MET-altered NSCLC remained a largely untapped segment, dependent on non-selective inhibitors such as crizotinib, which offered limited efficacy and lacked dedicated approvals.
Japan became the first country to approve a MET inhibitor therapy, tepotinib (TEPMETKO) (developed by EMD Serono [Merck KGaA]), on March 25, 2020. Following its initial approval in Japan, the FDA granted accelerated approval to tepotinib in the United States in February 2021.
On the other hand, developed by Novartis, capmatinib (TABRECTA) was the first MET inhibitor specifically approved by the US FDA on May 6, 2020, followed by tepotinib (TEPMETKO). Savolitinib (ORPATHYS) gained approval in China, and telisotuzumab vedotin (EMRELIS) was approved in May 2025 for high c-MET overexpression in nonsquamous NSCLC.
While NSCLC drugs like crizotinib and cabozantinib were approved earlier, they are multi-kinase inhibitors that target MET alongside other targets (like ALK or VEGFR), whereas capmatinib, tepotinib, etc., are highly selective for MET.
Novartis has strengthened its foothold in the METex14+ NSCLC market with capmatinib (TABRECTA), securing FDA approval in August 2022 for metastatic NSCLC with METex14 skipping mutations, following earlier nods in Europe (June 2022) and Japan (June 2020). These approvals mark a significant advancement in oncology, offering patients with METex14 mutations a targeted therapy option for improved outcomes.
The pivotal Phase II (GEOMETRY Mono-1) trial demonstrated Capmatinib (TABRECTA)’s efficacy, with an ORR of 68%, a PFS of 12.4 months, a DoR of 16.6 months, and an OS of 25.4 months in treatment-naïve patients. For previously treated patients, the ORR was 44%, with a PFS of 5.4 months and a DoR of 9.7 months. Safety data revealed dose reductions in 26% and dose interruptions in 57% of patients due to adverse events, emphasizing the need for effective management. TABRECTA’s strong clinical performance and global availability solidify its role as a key player in the competitive MET-targeted therapy landscape.
| Regulatory Landscape of c-MET Inhibitors TKI (Approved C-MET Inhibitors TKIs) among the 7MM + China | ||||||
| Drug Name | Company | Patient segment | Pivotal Study ID | Efficacy | Safety | Cost of Therapy |
| Savolitinib (ORPATHYS) | HutchMed & AstraZeneca | Locally advanced/metastatic NSCLC with METex14 skipping alteration | NCT04923945 | Treatment-Naïve ORR: 62.1%; PFS: 13.7 monthsOS: Not reached (at 20.8-month follow-up) | Common Grade 3+ Adverse Events (≥5%): Abnormal hepatic function (16.9%); Increased ALT (14.5%); Increased AST (12.0%) | ~USD 100,000 (considered mPFS: 7.2 months) |
| Previously TreatedORR: 39.2%PFS: 11.0 monthsmOS: Not mature | ||||||
| Capmatinib (TABRECTA) | Novartis | METex14 mutation-positive advanced and/or recurrent unresectable NSCLC | NCT02414139 (Geometry Mono-1) | Treatment NaïveORR: 68%;mDoR: 16.6 month;mPFS: 12;mOS: 25 | Permanent discontinuation of TABRECTA due to an adverse reaction occurred in 17% of patients. The most frequent adverse reactions (≥ 1%) leading to permanent discontinuation of TABRECTA were ILD/pneumonitis | ~USD 210,000 (considered mPFS: 9.7 months) |
| Previously TreatedORR: 44%; mDoR: 9.7 month;2L: mPFS: 6.93; mOS: 242L/3L: mPFS: 5.4; mOS: 13.57 | ||||||
| Tepotinib (TEPMETKO) | Merck KGaA | NSCLC w/MET Ex14 skipping | NCT02864992 (VISION) | Treatment-Naïve ORR: 57%;DoR: 1.3+ monthsPreviously Treated ORR: 45%;DoR: 1.4+ months | Serious adverse events occurred in 51% of patients; 25% of patients discontinued treatment due to adverse reactions | ~USD 220,000 (considered mPFS: 11 months) |
| Gumarontinib (HAIYITAN) | Haihe Biopharma & Taiho Pharmaceutical | NSCLC w/METex14 skipping | NCT04270591(GLORY) | For the overall population:ORR = 65.8%; PFS = 8.5 months; OS = 17.3 months | Grade ≥3 TRAEs: 53.6%Discontinuations due to TRAEs: 8.3% | ~USD 50,000 (considered mPFS: 11.7 month) |
| Telisotuzumab vedotin (EMRELIS) | AbbVie | c-MET protein overexpression | NCT03539536 (LUMINOSITY) | ORR: 35%; mDoR: 7.2 months | Common AEs (≥20%); Grade ≥3 labs (≥2%) | ~USD 400,000 (considered 15–16 vials) |
| Note: For HAIYITAN, the Japan price is considered, as the drug is approved in Japan and China; for all other drugs, US pricing has been used | ||||||
Evolution of the MET Landscape: Transition from MET Inhibitors to MET-targeted ADCs
Teliso-V (telisotuzumab vedotin; EMRELIS) has secured FDA accelerated approval as the first therapy for cMet-overexpressing NSCLC, establishing a new treatment avenue. However, its use is confined to a limited subset, patients with high cMet expression (≥50% tumor cells with IHC 3+), effectively narrowing its reach to a niche population.
The approval is driven by results from the LUMINOSITY trial, where efficacy was most pronounced in patients with the highest levels of cMet expression. While ~25% of EGFR wild-type, non-squamous NSCLC patients exhibit cMet overexpression, only about half meet the high-expression threshold required for treatment eligibility. Despite this milestone, safety considerations remain, including a warning for interstitial lung disease (ILDs), and strategic focus appears to be shifting toward next-generation assets.
AbbVie has discontinued plans to expand Teliso-V in post-osimertinib (TAGRISSO) settings, instead advancing teliso-A combinations, including a Phase II/III first-line trial with osimertinib that does not restrict enrollment based on cMET expression levels, signaling a shift toward more inclusive and potentially scalable strategies. Telisotuzumab adizutecan (teliso-A) is built on the same antibody backbone but with a more potent topoisomerase I inhibitor payload (vs. MMAE in Telisotuzumab vedotin). Telisotuzumab vedotin (EMRELIS) is emerging as a key successor in cMet NSCLC. Early clinical data from a Phase I basket study presented at the ASCO Annual Meeting suggest promising activity for teliso-A, with ORR reaching 63% in heavily pretreated EGFRm NSCLC patients, including those with lower cMet expression, highlighting its broader potential. However, these findings remain preliminary and require validation in larger, controlled trials.
| Clinical Trial Overview of AbbVie c-MET–Targeted Therapies in NSCLC | |||
| Trial | Setting | Design | Note |
| Temab-A (Telisotuzumab adizutecan) | |||
| Phase II/III (M25-287) | 1L EGFR metastatic non- squamous NSCLC | Osimertinib (TAGRISSO) combo vs. Osimertinib (TAGRISSO) | cMET status must be available; started August 2025 |
| Phase II/III (M25-713) | 2L EGFR metastatic non-squamous NSCLC (post-3rd gen TKI) | Monotherapy treatment vs. the investigator’s choice | No mention of cMET status |
| Telisotuzumab vedotin (EMRELIS) | |||
| Phase II(Luminosity) | 2L EGFRwt cMET +ve NSCLC | Monotherapy treatment uncontrolled | Basis for US accelerated approval as EMRELIS, in cMET ≥50% with IHC 3+ |
| Phase III (Telimet NSCLC-01) | 2L EGFRwt cMET + ve (high and intermediate) NSCLC | Monotherapy treatment vs. chemo | Confirmatory study |
| Phase III (M22-142) | 2L EGFRm cMET + ve NSCLC | Osimertinib (TAGRISSO) combo vs. chemotherapy | Withdrawn for “strategic considerations.” |
C-MET NSCLC 2026+: Mapping the Upcoming Competitive Arena
The NSCLC pipeline has evolved into a competitive landscape. Advances around the MET signaling pathway have driven a surge in innovation, with next-generation MET inhibitors aiming to overcome resistance and improve durability.
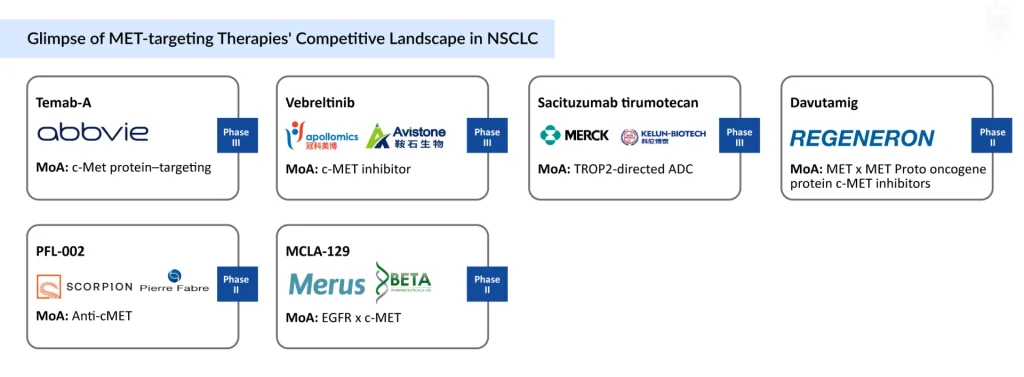
At the same time, combination strategies are gaining momentum, particularly MET with EGFR signaling pathway inhibitors. The Phase III (SAFFRON) trial evaluating Savolitinib (ORPATHYS) + Osimertinib (TAGRISSO) highlights this shift toward biology-driven approaches.
Innovation is also expanding beyond small molecules, with novel modalities like ADCs. Merus/Betta Pharmaceuticals’ MCLA-129 is an ADCC-enhanced human IgG1 Biclonics that targets both EGFR and c-MET. This NSCLC drug is currently being evaluated in Phase I/II trials as a monotherapy for locally advanced or metastatic c-METex14 skipping NSCLC.
Conclusion
The adoption of NGS has established MET exon14 skipping as a distinct, actionable driver in NSCLC, enabling targeted therapies such as capmatinib and tepotinib to deliver meaningful clinical benefit. To maximize impact, strategies should integrate standardized diagnostics, biomarker‑driven patient selection, rational combinations, especially with EGFR inhibitors, next‑generation MET inhibitors, and novel modalities like ADCs and bispecific antibodies, with a focus on early‑line use and enriched NSCLC trial designs.
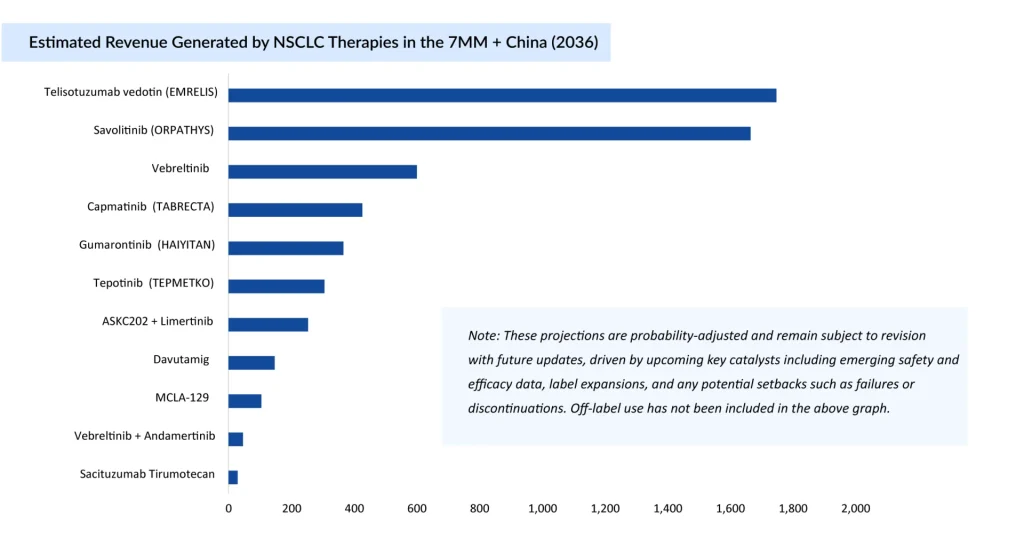
The journey of MET‑NSCLC reflects a shift from a neglected, poorly defined niche to a recognized, competitive, and increasingly promising segment. Advances in NGS, biomarker identification, and targeted agents, driven in part by EGFR TKI resistance, have repositioned MET‑NSCLC as a high‑priority frontier where combination regimens, refined biomarkers, and innovative modalities are likely to shape long‑term outcomes.

Downloads
Article in PDF
Recent Articles
- Merck’s Keytruda sales; Valeant on name change; Pfizer – BMS; Amgen puts Repatha outcomes for deal
- Takeda and AC Immune’s Alzheimer’s Deal; Eli Lilly’s Donanemab FDA Review; Bristol Myers Sq...
- Non-Small Cell Lung Cancer Market: Treatments and Market Forecast
- Daiichi Sankyo’s Trastuzumab Deruxtecan; ODD to Bexmarilimab for AML; Roche’ Alecensa; Ergomed Ai...
- Evotec, Takeda Deal; AZ COVID Vaccine US Trials; Promega’s Recognition; Roche’s Tecentriq R...