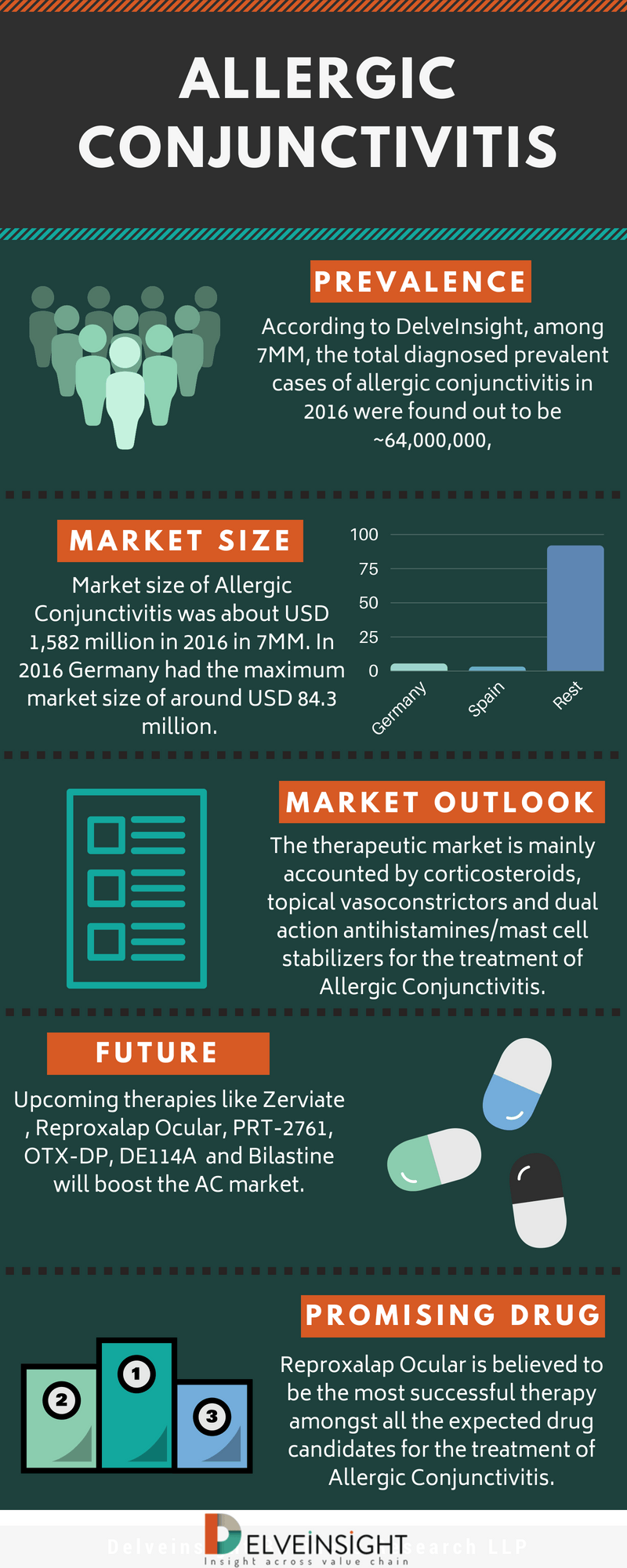
Allergic Conjunctivitis (AC)- Market Insights, Epidemiology and Market Forecast- 2027
Jul 10, 2018
Downloads
Click Here To Get the Article in PDF
Downloads
Article in PDF
Recent Articles
- AstraZeneca and Incyte Collaborate; Tocagen’s therapy; FDA warns companies;FDA Revokes
- Business Cocktail
- Cervical Cancer: Current Scenario
- Understanding the Acute Myeloid Leukemia Surge and the Road to Solutions
- SEC scrutinizes; Acacia meets endpoint; Axovant’s dementia drug; Novartis expands; Aurobind...