5 Emerging Polycythemia Vera Therapies Eyeing Market Dominance
Apr 10, 2026
Summary
- Three drugs that have been approved for PV include JAKAFI/JAKAVI (ruxolitinib), BESREMi (ropeginterferon alfa-2B/AOP2014/P1101), and PEGASYS (peginterferon alfa-2a). JAKAFI and BESREMi are approved in the US, Europe, and Japan, while PEGASYS is approved only in Europe.
- JAKAFI is nearing its patent expiry in 2028; however, as of now, its revenue is increasing continuously due to stronger-than-expected demand, particularly from patients with polycythemia vera and graft-versus-host disease.
- Key players involved in developing potential therapies for the treatment of PV include Protagonist Therapeutics and Takeda (Rusfertide), Italfarmaco (DUVYZAT), Ionis Pharmaceuticals and Ono Pharmaceuticals (Sapablursen), Merck (Imago BioSciences) (Bomedemstat), Disc Medicine (DISC-3405), and others.
Polycythemia vera treatment in the US is at a turning point. Despite decades of management strategies, the disease still has no curative therapy, and treatment options remain limited. For years, the landscape was largely shaped by legacy therapies such as hydroxyurea, second-generation interferons, and phlebotomy. This long-standing paradigm began to shift with the approval of PharmaEssentia/AOP Orphan Pharmaceuticals’ BESREMi, the first interferon specifically approved for polycythemia vera treatment and now used in both first- and second-line settings.
Meanwhile, JAKAFI continues to compete in the second-line space alongside platelet-reducing agents such as anagrelide, interferon-based therapies, and BESREMi, while options beyond second line remain scarce. Although JAKAFI is approaching patent expiry in 2028, its revenues continue to climb, driven by strong demand from patients with polycythemia vera and graft-versus-host disease.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- FDA Approves Eisai & Biogen’s LEQEMBI IQLIK for Maintenance Treatment of Early Alzheimer’s; T...
- JAK Inhibitors: New Lifeline for Hair Loss Treatment
- Essential Thrombocythemia Treatment—Why the US Lags Behind Europe?
- Madrigal Wins EU Approval for REZDIFFRA in MASH With Liver Fibrosis; Valneva Faces FDA License Su...
- Assessment of Key Products that Got FDA Approval in Second Half (H2) of 2021
Interferons have long played a role in disease management. While BESREMi is the first interferon formally approved for Polycythemia Vera, physicians have historically relied on PEGASYS, a pegylated interferon widely used off-label in patients with Myeloproliferative Neoplasms. However, off-label use has often created access and reimbursement challenges for patients.
Current PV treatment strategies still follow a risk-adapted approach. Patients with low thrombotic risk typically receive low-dose aspirin and phlebotomy, whereas cytoreductive therapy, most commonly hydroxyurea or interferon-alpha, is recommended for high-risk patients. Yet the need for better therapies remains pressing: around 30% of patients treated with hydroxyurea develop resistance or intolerance, leaving limited alternatives.
Currently, there are no effective strategies for the reduction of the transformation of PV to myelofibrosis, which can occur in 10–20% of patients, and transformation to acute myeloid leukemia, which can occur in another 5–10% of patients, as per Sadaf Javed, Manager of Forecasting at DelveInsight. There is a need for treatment options that can decrease the risk of transformation to either myelofibrosis or acute leukemia, according to Javed.
Javed also stated that the limitations of pharmacological treatment methods are one of the major concerns for polycythemia vera patients. This situation, Javed said, is concerning as PV is a rare disease affecting older people, and proper management can lead to other heart-related issues.
The future, however, looks far more dynamic. The next wave of innovation is expected to reshape the polycythemia vera drug landscape, with emerging modalities including iron modulators, LSD1 inhibitors, pan-HDAC inhibitors, TMPRSS6 inhibitors, transferrin receptor (TfR) targeting agents, BCL-2 inhibitors, immunostimulants, CK1α modulators, and several other novel mechanisms, potentially ushering in a new era of precision-driven treatment options for polycythemia vera.
Here, DelveInsight spotlights the 5 most promising emerging therapies, innovative contenders that could disrupt the market leaders if approved.
Protagonist and Takeda’s Rusfertide Driving a Hepcidin Mimetic Breakthrough in Phlebotomy Control
Leading the pack is rusfertide, a next-generation injectable synthetic mimetic of the natural hormone hepcidin, engineered for superior potency, solubility, and stability, resulting in improved in vivo pharmacokinetics, pharmacodynamics, and manufacturability compared with the native hormone.
Momentum around rusfertide continues to build. At the American Society of Hematology Annual Meeting in December 2024, final results from the Phase II REVIVE trial demonstrated that adding rusfertide to therapeutic phlebotomy, with or without cytoreductive therapy, effectively controlled erythrocytosis, delivered durable hematocrit control, and significantly reduced the need for phlebotomy in patients with polycythemia vera.
Reflecting its therapeutic promise, the FDA granted rusfertide orphan drug, fast track, and breakthrough therapy designations for this indication. The therapy is now being evaluated in the pivotal Phase III VERIFY trial, while its developer, Protagonist Therapeutics, is simultaneously advancing an oral hepcidin formulation currently in the preclinical stage, potentially expanding the treatment paradigm even further.
Clinical momentum accelerated in March 2025 when Protagonist Therapeutics and Takeda Pharmaceutical Company announced positive topline results from the Phase III VERIFY study. The trial, which evaluated rusfertide as an add-on to standard of care in phlebotomy-dependent patients with polycythemia vera, successfully met its primary endpoint and all four key secondary endpoints, reinforcing the drug’s potential to reshape disease management.
Further insights were presented at the 67th Annual ASH Meeting in December 2025, where additional clinical data on rusfertide, including findings from the Phase II/III VERIFY program, are scheduled for presentation. With the FDA accepting the NDA and granting priority review, the therapy is now edging closer to market, with an anticipated launch window in the third quarter of 2026.
According to Aparna Thakur, Assistant Project Manager of Forecasting, DelveInsight, rusfertide is poised to be the first among emerging therapies to enter the polycythemia vera market, giving it a clear first-mover advantage. Thakur further said that with the NDA already submitted and a launch expected this year, rusfertide is well-positioned to move earlier in the treatment paradigm and has the potential to meaningfully reshape the management of polycythemia vera.
Italfarmaco’s DUVYZAT Targeting High-Risk JAK2-Positive PV Through HDAC Inhibition
Givinostat, an orally bioavailable hydroxamate histone deacetylase (HDAC) inhibitor, is emerging as a promising therapy with anti-inflammatory, anti-angiogenic, and antineoplastic potential. The drug achieved a major milestone in March 2024 with its first regulatory approval for the treatment of Duchenne muscular dystrophy, marketed under the brand name DUVYZAT.
Earlier, in September 2017, the FDA granted Orphan Drug Designation to Italfarmaco for givinostat to treat Polycythemia Vera. Building on this momentum, the company is currently advancing the therapy through Phase III clinical trials for polycythemia vera, with a potential launch anticipated by 2027.
Further strengthening its regulatory pathway, in May 2025, the FDA also awarded Fast Track designation to givinostat for patients with polycythemia vera, an area where effective treatment options remain limited.
Givinostat could emerge as a next-generation first-line cytoreductive option, offering a well-tolerated alternative to hydroxyurea for long-term management of polycythemia vera, as per Thakur.
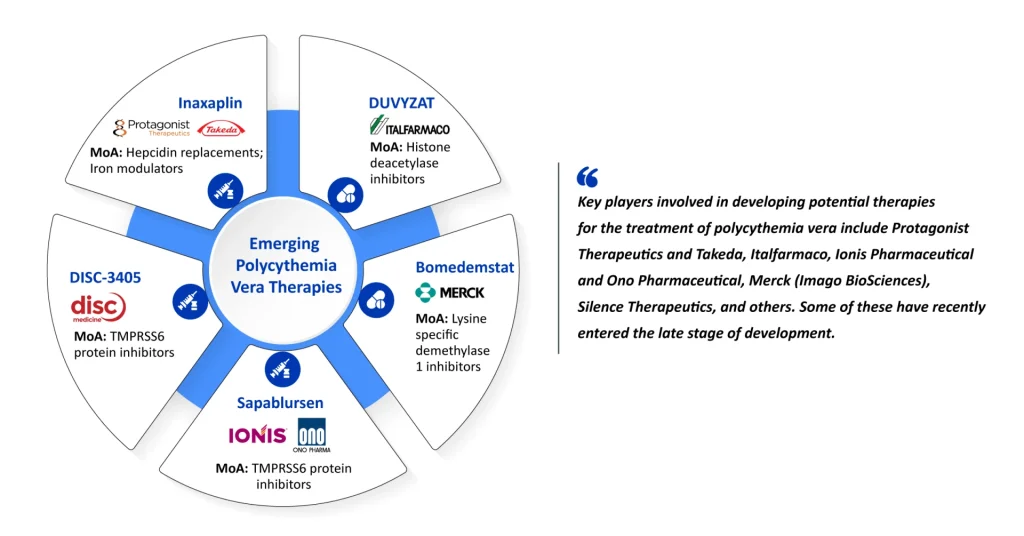
Merck’s Bomedemstat Targeting Stem Cells Through LSD1 Blockade
Bomedemstat is an orally bioavailable small-molecule inhibitor targeting lysine-specific demethylase 1 (LSD1/KDM1A), a key epigenetic regulator critical for the survival and proliferation of cancer stem and progenitor cells, particularly malignant bone marrow cells. The therapy is currently being investigated in a Phase III clinical trial (NCT06351631) for polycythemia vera, while also being explored as a potential treatment option for myelofibrosis.
Although polycythemia vera remains a secondary development focus at present, successful clinical outcomes in essential thrombocythemia could pave the way for expanding bomedemstat’s therapeutic scope into the polycythemia vera landscape, positioning the drug as a promising candidate in the broader myeloproliferative neoplasm (MPN) treatment space.
Adding further momentum, in November 2025, Merck & Co. unveiled the new data spanning multiple hematologic malignancies at the American Society of Hematology Annual Meeting and Exposition in December 2025. The presentations spotlight key pipeline candidates, including bomedemstat (MK-3543), highlighting Merck’s continued commitment to advancing innovative therapies for hematologic cancers.
Javed stated that bomedemstat is best viewed as a later-line or second-wave entrant in the polycythemia vera landscape, with regulatory submission and commercial launch not expected before 2029, contingent on positive Phase III outcomes and clear differentiation versus existing standards of care.
Ionis and Ono’s Sapablursen Bringing Antisense Precision to the Iron Axis
Sapablursen is engineered to target TMPRSS6, unlocking increased production of hepcidin, the master regulator of iron balance in the body. By restoring hepcidin levels, sapablursen aims to correct iron dysregulation and potentially transform the treatment landscape for blood disorders such as polycythemia vera (PV).
Clinical momentum is growing. In December 2025, Deciphera Pharmaceuticals, a member of Ono Pharmaceutical Co., Ltd., announced the oral presentation of encouraging Phase 2a IMPRSSION study results for sapablursen in patients with PV at the 67th American Society of Hematology Annual Meeting. The study was conducted by Ionis Pharmaceuticals, which originally discovered and developed the therapy.
Earlier in March 2025, Ionis and Ono entered into a licensing agreement granting Ono exclusive global rights to develop and commercialize sapablursen. The therapy is currently being evaluated in adults with PV in the fully enrolled Phase 2 IMPRSSION trial, highlighting its advancing polycythemia vera clinical progress.
Notably, sapablursen also received both Fast Track and Orphan Drug designations from the FDA in 2024, underscoring its potential as a promising new treatment option for patients living with PV.
Disc Medicine’s DISC-3405 Offers Precision Antibody Targeting of TMPRSS6
DISC-3405 is an investigational monoclonal antibody designed to target TMPRSS6 (Transmembrane Serine Protease 6, also known as Matriptase-2), triggering the body’s natural production of hepcidin. By elevating hepcidin levels and reducing iron availability, the therapy aims to address a broad spectrum of diseases driven by iron dysregulation.
Originally in-licensed from Mabwell Therapeutics, DISC-3405 (formerly MWTX-003) has already demonstrated encouraging progress, with a Phase 1 trial completed in healthy volunteers. The program has now advanced into a Phase 2 study in polycythemia vera and a Phase 1b trial in sickle cell disease, with further exploration planned across additional iron overload disorders.
As these novel polycythemia vera candidates advance through late-stage development and approach potential commercialization, they hold the promise not only to improve hematocrit control and reduce treatment burden but also to fundamentally alter disease trajectory. Collectively, these innovations signal the dawn of a new era in polycythemia vera management, one that prioritizes long-term disease control, improved patient outcomes, and, ultimately, the possibility of disease modification.

Downloads
Article in PDF
Recent Articles
- JAK Inhibitors: New Lifeline for Hair Loss Treatment
- LEQEMBI Intravenous Infusion Approval; Novartis’ Presented Updates on Lutathera; FDA Accept...
- Madrigal Wins EU Approval for REZDIFFRA in MASH With Liver Fibrosis; Valneva Faces FDA License Su...
- Polycythemia Vera: Growing Competition and the Changing Treatment Landscape in 2025
- The Dynamic Landscape of Myelofibrosis Treatment: A 2024 Perspective