Unlocking Growth Potential in the Presbyopia Treatment Market: Innovations Reshaping Age-Related Vision Loss
Apr 06, 2026
Table of Contents
Summary
- According to DelveInsight’s analysis, in 2025, the total prevalent cases of presbyopia were approximately 280.5 million cases in the 7MM. The total number of diagnosed cases of presbyopia was approximately 235 million cases.
- A significant proportion of presbyopia patients, nearly 65%, continue to rely on eyeglasses for near vision correction, with spectacles accounting for roughly half of the US market revenue as of 2025.
- The development landscape is gaining momentum, with key players such as Opus Genetics/Viatris and Eyenovia advancing late-stage candidates, while additional contenders like Glaukos Corporation are progressing mid-stage therapies, intensifying competition.
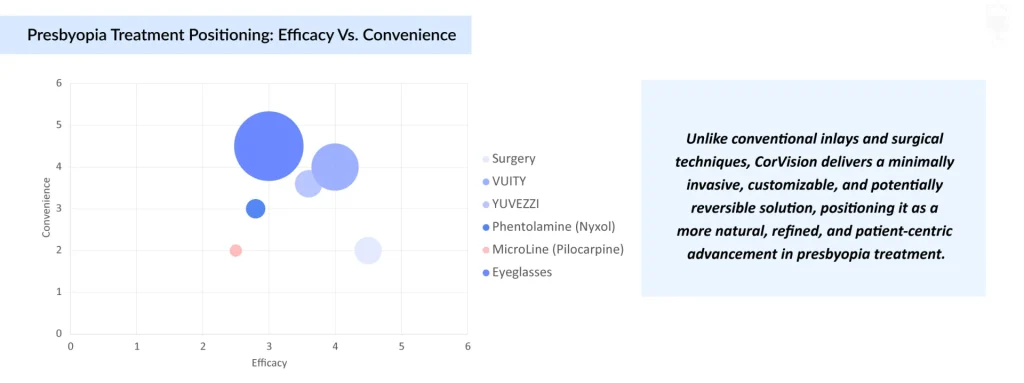
- The approval of VUITY has set a clear benchmark as the first approved presbyopia eye drop, demonstrating patients gaining ≥3 lines in near visual acuity (mesopic, high-contrast binocular DCNVA) without clinically meaningful loss in distance vision and convenient once-daily dosing.
- This transition signals a shift toward branded, premium pharmacological solutions, creating new revenue opportunities for pharma players while gradually reducing dependence on commoditized optical correction.
- Building on this momentum, newer entrants like YUVEZZI are poised to strengthen competition by leveraging increased awareness and offering differentiated formulations, positioning themselves to capture within the growing pharmacologic segment.
- According to DelveInsight’s estimates, the total market size of presbyopia in the 7MM was close to USD 17,000 million in 2022 and is projected to grow over the forecast period (2026–2036), driven by the anticipated launch of emerging therapies, such as Phentolamine.
- The primary unmet needs, such as inconsistent access to affordable and effective presbyopia correction, limitations of current treatment options, and lack of personalized solutions, represent major barriers. Addressing these issues would facilitate market expansion and improve disease management.
Presbyopia is an age-driven visual condition that gradually diminishes the eye’s ability to focus on nearby objects, typically emerging after the age of 40. The condition stems from progressive structural and functional changes within the eye, particularly involving the crystalline lens and its supporting apparatus. Once flexible and responsive, the lens becomes increasingly rigid over time, impairing the eye’s accommodative capacity. This transformation reflects a broader ocular aging process, making presbyopia not merely a refractive issue but a hallmark of physiological aging.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Contact Lenses: A Comprehensive Guide to Vision Correction and Beyond
- How Pharmaceutical Companies Are Mitigating The Gap In The Eye Disorders Treatment Market
- How will Pharmacotherapies help the growing number of Presbyopes?
- LumiThera’s US LIGHTSITE III Trial; First Patient Enrolled in Vascular Closure Device Study of Te...
- Novo Nordisk A/S Announces that CagriSema Delivered Superior Outcomes in REIMAGINE 2; Sanofi’s Ve...
Clinically, presbyopia manifests as a gradual decline in near vision, often first noticed during reading or other close-up tasks, especially under low-light conditions or prolonged use. Individuals may experience eye strain, headaches, and the need to hold objects at arm’s length to achieve clarity. While distance vision generally remains unaffected, the burden on daily activities and productivity can be substantial. With over a billion people affected worldwide, the condition has driven growing interest in innovative presbyopia management strategies, ranging from corrective lenses to emerging pharmacological and surgical interventions.
“Topical drops for treating presbyopia are a whole new class of therapeutics that will explode over the next few years. Ophthalmologists need to embrace these medications, which look very promising in clinical trials.”
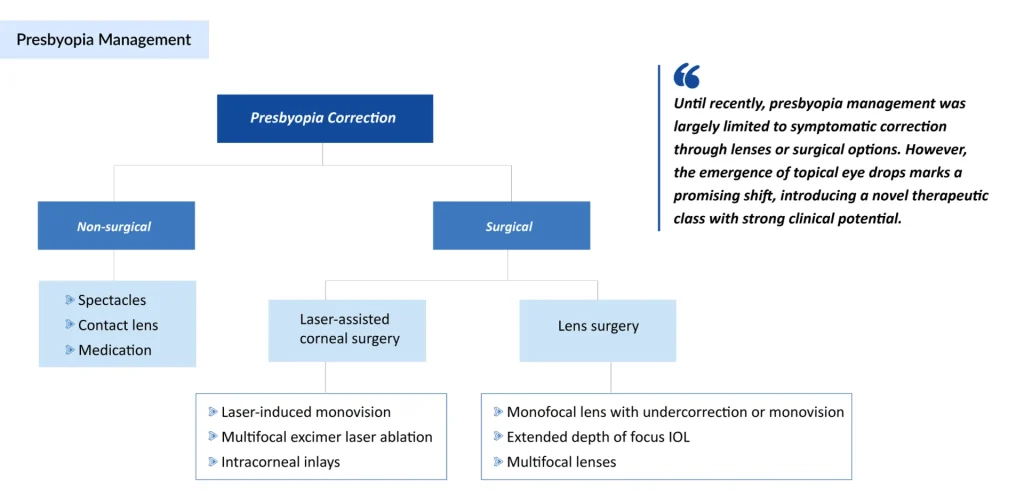
The management of presbyopia has evolved into a dynamic, patient-centric approach aimed at restoring near vision without compromising distance clarity. Traditional optical solutions such as spectacles and contact lenses remain widely used, offering flexible and cost-effective correction options. Meanwhile, pharmacological therapies, particularly miotic agents, are emerging as innovative, noninvasive alternatives that enhance depth of focus. On the surgical front, advanced corneal procedures, intracorneal inlays, and lens-based interventions, including EDOF and accommodating intraocular lenses, are expanding the therapeutic arsenal. Together, these options underscore a rapidly advancing landscape where personalized treatment strategies are key to optimizing visual outcomes and quality of life.
In October 2021, the US FDA approved VUITY (pilocarpine HCl ophthalmic solution) 1.25%, developed by Allergan, an AbbVie company, as the first pharmacological treatment for presbyopia in adults. In the US, this topical therapy is indicated for age-related blurry near vision and is administered as once-daily eye drops to improve near visual acuity.
Recently, in January 2026, Tenpoint Therapeutics, Ltd. announced that the FDA approved YUVEZZI (carbachol and brimonidine tartrate ophthalmic solution) 2.75%/0.1%, previously known as BRIMOCHOL PF, the first and only dual-agent eye drop for the treatment of presbyopia in adults.
Presbyopia Treatment Challenges
High costs associated with corrective lenses, surgical procedures, and ongoing eye care act as a major barrier to presbyopia treatment. These expenses limit affordability, particularly in cost-sensitive populations, leading to lower adoption rates of available treatment options.
Socioeconomic barriers, poor eye health literacy, and limited awareness about available treatments often delay diagnosis and reduce utilization of presbyopia management options. As a result, many individuals postpone or avoid proper correction, ultimately impacting their quality of vision and daily functioning.
Emerging Therapies to Improve Presbyopia Treatment
Emerging presbyopia therapies such as Phentolamine are advancing novel pharmacological approaches that improve near vision through targeted pupil modulation, offering differentiated, non-invasive treatment options.

Adding to this momentum, early-stage innovation continues to expand the landscape, with TPT-161 and CBT-199 currently in preclinical development, exploring new mechanisms to enhance visual outcomes and patient convenience. Collectively, these therapies underscore a rapidly progressing pipeline poised to redefine presbyopia management with more effective, patient-friendly solutions.
Opus Genetics/Viatris’ Phentolamine
Phentolamine Ophthalmic Solution 0.75%, developed by Opus Genetics, is a late-stage investigational therapy for presbyopia that works as a non-selective alpha-adrenergic antagonist. Modulating pupil size through inhibition of iris dilator muscle activity, it enhances near vision without directly impacting ciliary muscle function. The therapy is currently under regulatory review following submission of an sNDA in early 2026.
What sets this therapy apart is its non-miotic mechanism and potential for consistent visual improvement without compromising distance vision, a common limitation with existing options. Supported by positive Phase III results, it offers a promising safety profile and convenient dosing, positioning it as a differentiated, next-generation pharmacological approach in the evolving presbyopia landscape, with Opus Genetics anticipating a regulatory decision by the end of 2026.
Emerging Medical Devices to Improve Presbyopia Treatment
Emerging ophthalmological medical devices are redefining presbyopia management by introducing innovative, non-traditional approaches beyond glasses and drugs. CorVision®, developed by Linkcore, is a minimally invasive implant designed to improve near vision in presbyopic adults, while the riboflavin/UV-A-based phototherapy device from RegenSight is in early prototyping stages, exploring corneal remodeling techniques. Together, these advancements signal a shift toward next-generation, device-driven solutions aimed at enhancing visual outcomes with improved patient comfort and flexibility.
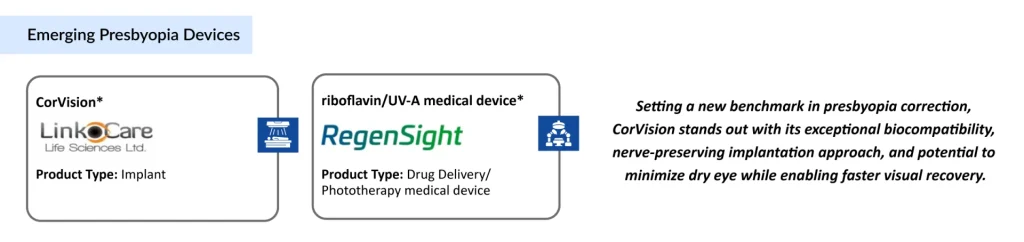
LinkoCare Life Sciences’ CorVision
CorVision, developed by Linkcore, is a next-generation bioengineered corneal microlens designed for presbyopia management. Made from collagen, the same natural material as the human cornea, it is implanted within a corneal pocket, enabling seamless integration while minimizing risks associated with traditional corneal inlays and more invasive procedures.
Unmet Needs in Presbyopia Treatment
Despite being easily diagnosable and treatable, presbyopia continues to face substantial gaps in access and awareness, with nearly half of affected individuals worldwide lacking adequate correction, particularly in low- and middle-income regions due to limited screening, high costs, and poor access to vision care. Even in developed markets, it is often overlooked as a natural part of aging, resulting in delayed intervention and reduced productivity.
This disparity creates a clear opportunity to expand scalable, low-cost vision solutions and strengthen primary eye care systems through integrated screening, improved awareness, and subsidized correction. Overcoming these barriers can drive earlier diagnosis and presbyopia treatment while enabling the development of cost-effective models tailored to underserved markets, ultimately enhancing quality of life and advancing global visual health equity.
Beyond access, the under-recognition of presbyopia’s real-world impact further constrains effective management. Clinical evaluations often overlook functional limitations, digital strain, and psychosocial effects, while adaptive behaviors delay diagnosis, resulting in suboptimal, non-personalized care and reduced daily performance.
This underscores the need for a shift toward a more patient-centered approach, integrating functional vision assessments and validated patient-reported outcome measures into routine care. Such an approach would enable more tailored interventions, while therapies demonstrating improvements in daily functioning, productivity, and digital comfort can achieve stronger differentiation and real-world value recognition.
Presbyopia Treatment Market Outlook for the Future
The presbyopia market is expected to witness steady growth in the coming years, driven by a combination of surgical, non-surgical, and pharmacological treatment options. Advanced procedures such as refractive surgery, lens implants, and corneal inlays will continue to gain adoption due to their long-term vision correction benefits. At the same time, optical solutions like spectacles and contact lenses will remain widely used due to their affordability and accessibility. Increasing awareness and early diagnosis are also likely to support market expansion across diverse populations.
The introduction of pharmacological therapies is set to transform the presbyopia market, offering a less invasive alternative to traditional treatments. The approval of eye drops such as VUITY and newer options like YUVEZZI highlights growing interest in drug-based solutions. In addition, several investigational therapies are under development, which are expected to further expand treatment possibilities. These innovations are likely to improve patient compliance and accessibility, especially among those seeking non-surgical options. Overall, pharmacological advancements are poised to play a key role in shaping the future growth and direction of the presbyopia market.
Barriers to Adoption
While the emergence of pharmacological therapies marks a meaningful shift, adoption is likely to be moderated by several real-world barriers. Cost and reimbursement limitations remain key challenges, particularly in price-sensitive markets where out-of-pocket spending dominates, and lower-cost optical correction continues to be the default choice. For instance, with eyeglasses costing approximately USD 100 annually compared to around USD 700 for premium pharmacologic options like VUITY, the price differential may deter widespread adoption. Furthermore, the variability in patient response, the need for consistent daily dosing, and concerns around side effects such as headaches or dim vision in low-light conditions may impact long-term adherence. Physician inertia and limited familiarity with newer therapies could further slow uptake in routine clinical practice.
Moreover, the perceived value proposition of these treatments must compete with well-established, low-cost alternatives like reading glasses and contact lenses. In many cases, presbyopia is still viewed as a manageable inconvenience rather than a condition requiring medical intervention, which can limit willingness to adopt premium therapies. The positioning of pharmacologic treatments within a mid-tier pricing bracket between inexpensive optical correction and high-cost surgical interventions may also create hesitation among consumers evaluating cost versus convenience.
A similar dynamic is observed in the myopia medical devices landscape, where traditional corrective lenses continue to dominate due to their accessibility, even as advanced technologies such as orthokeratology lenses and AI-enabled diagnostic tools gain traction but face cost and adoption barriers. Regulatory and market access hurdles, along with the need for sustained patient education and evidence of real-world benefits, may continue to temper the pace of widespread adoption despite strong clinical promise.

Frequently Asked Questions
The current approved treatments for presbyopia primarily include pharmacological eye drops designed to improve near vision through pupil modulation. Key approvals include VUITY™ (pilocarpine HCl ophthalmic solution) 1.25% by AbbVie, QLOSI™ (pilocarpine hydrochloride ophthalmic solution) 0.4% by Orasis Pharmaceuticals, VIZZ (aceclidine ophthalmic solution) 1.44% by LENZ Therapeutics, and YUVEZZI™ (carbachol and brimonidine tartrate ophthalmic solution) by Tenpoint Therapeutics, marking a shift toward non-invasive, drop-based therapies.
However, these treatments come with limitations, including temporary effects requiring daily dosing, variable patient response, and potential side effects such as dim vision or headaches due to induced pupil constriction.
Emerging therapies for presbyopia are rapidly reshaping the treatment landscape, with innovative pharmacological approaches taking center stage. Candidates such as Phentolamine are advancing targeted pupil modulation strategies to enhance near vision, offering non-invasive and differentiated alternatives to current options. Alongside this, a strong late-stage pipeline, including therapies like MicroLine (pilocarpine ophthalmic) and LENZ101, reflects growing clinical progress. Further strengthening this momentum, early-stage candidates such as TPT-161 and CBT-199 are exploring novel mechanisms to improve visual outcomes and patient convenience.
Presbyopia treatment continues to face key unmet needs, including limited access to affordable and effective correction, particularly in underserved populations. Most current options provide only temporary, symptomatic relief without restoring natural accommodation. Additionally, patient-centric factors such as quality of life, digital strain, and daily functional impact are often underaddressed in clinical care. Delayed diagnosis and lack of proactive screening further hinder timely intervention and optimal management.
According to DelveInsight, in the “7MM” (major markets), the Presbyopia market was approximately USD 17,000 million in 2022. The market is projected to grow at a Compound Annual Growth Rate (CAGR) of about 4% from 2026 through 2036. The growth of the presbyopia market is driven by emerging therapies, increasing awareness, and earlier diagnosis, alongside continuous advancements in treatment options. These factors are expanding access to care and supporting the overall rise in market size.
Future transformations in presbyopia therapy and patient care will be driven by the increasing proportion of middle-aged and elderly individuals, as age-related near vision decline continues to rise globally. Continuous advancements in corrective lenses, surgical techniques, and intraocular lens technologies, along with emerging pharmacological therapies, are expanding and refining treatment options. Ongoing research into presbyopia-correcting eye drops and other non-surgical approaches is expected to introduce more convenient and less invasive alternatives. Together, these developments will enable more personalized, accessible, and effective management of presbyopia, reshaping both treatment strategies and patient care outcomes.
Downloads
Article in PDF
Recent Articles
- Novo Nordisk A/S Announces that CagriSema Delivered Superior Outcomes in REIMAGINE 2; Sanofi’s Ve...
- Alcon’s 510(k) Clearance From the FDA; Globus Medical Received FDA Clearance; UK Pioneers F...
- Insights Into the Evolving Landscape of the Ocular Implants Market
- How will Pharmacotherapies help the growing number of Presbyopes?
- Olympus Secures FDA Clearance for POWERSEAL™ Open Extended Jaw Device; Abbott’s Ultreon™ 3.0 Rece...




