Bone Marrow Failure: The Silent Global Crisis Driving a New Era of Therapeutic Innovation
Feb 23, 2026
Table of Contents
Summary
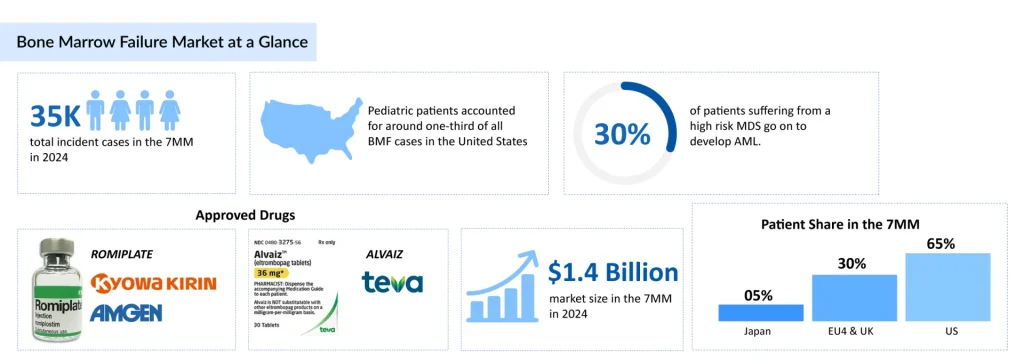
- In 2024, the 7MM reported approximately 35,000 incident BMF cases, highlighting the persistent clinical burden and the ongoing need for earlier diagnosis and more effective treatment strategies.
- ROMIPLATE has made notable regulatory progress in Japan, its only approved market for aplastic anemia, expanding from a restricted indication for refractory patients in 2019 to full approval for all aplastic anemia cases in 2023, reinforcing its increasing clinical importance within the country’s therapeutic landscape.
- Teva’s 2023 FDA approval of ALVAIZ for adults with severe aplastic anemia unresponsive to immunosuppressive therapy expands treatment options for a historically underserved population and introduces meaningful competition within the TPO-receptor agonist class.
- Emerging BMF therapies, including CK0801 (Cellenkos), EXG-34217 (Elixirgen Therapeutics), sonrotoclax [BGB-11417; BeOne Medicines (SHA: 688235)], and bexmarilimab + SoC [(Faron Pharmaceuticals (LON: FARN)] combined with standard of care, represent a new wave of mechanism-focused innovation addressing immune dysfunction, telomere maintenance, apoptotic pathways, and macrophage activity.
- The BMF market across the 7MM reached an estimated USD 1.4 billion in 2024, reflecting both the sustained clinical need and the continued reliance on established therapeutic approaches, even as early-stage innovation begins to reshape future care pathways.
Bone marrow failure may sound rare, but its impact is anything but small. Globally, bone marrow failure syndromes affect tens of thousands each year, with incidence rising alongside improved diagnostic capabilities and aging populations. According to global health trends reported by the World Health Organization, conditions like aplastic anemia occur in 2–6 people per million annually, with certain regions, particularly parts of Asia, experiencing rates up to three times higher.
Downloads
Click Here To Get the Article in PDF
In 2024, the 7MM recorded roughly 35K incident bone marrow failure cases, underscoring the steady clinical burden and the continued need for earlier diagnosis and effective treatment strategies. Based on DelveInsight estimates, pediatric patients accounted for around one-third of all BMF cases in the United States in 2024, with adults representing the dominant share at roughly 70%.
Despite advances in diagnostics and treatment, many patients still face delayed recognition because early symptoms, fatigue, frequent infections, and unexplained bruising are easy to overlook. As global populations age and environmental exposures shift, bone marrow failure syndromes are becoming an increasingly important public health concern.
Current Bone Marrow Failure Therapeutic Landscape
Stem cell transplantation remains the only definitive curative option for many bone marrow failure disorders, and its role is strengthening as the ecosystem around it evolves. Expanded transplant infrastructure, broader donor registries, and more refined conditioning regimens are enabling wider adoption, while newer approaches, such as haploidentical transplantation and cord blood–based procedures, are increasing the feasibility of transplant for patients who previously lacked suitable donor options.
Alongside this, thrombopoietin-receptor agonists have become firmly embedded in modern bone marrow failure treatment pathways, creating a therapeutic bridge for patients who are not immediate transplant candidates or who require adjunctive support. ROMIPLATE [Kyowa Kirin (TYO: 4151)/Amgen (NASDAQ: AMGN)], a recombinant TPO-receptor stimulator, exemplifies this evolution. Initially launched in Japan for ITP in 2011, it received an expanded approval for aplastic anemia patients unresponsive to conventional therapy in 2019, and in 2023, its indication was widened to include all aplastic anemia, reflecting its growing clinical integration.
Similarly, ALVAIZ [Teva Pharmaceuticals (TLV: TEVA)], which binds the transmembrane region of c-Mpl to promote megakaryocyte proliferation and platelet production, is reshaping the therapeutic landscape. Its 2023 authorization by the FDA for severe aplastic anemia treatment following IST introduces direct competitive momentum within the TPO-RA class. Together, these developments illustrate how supportive pharmacologic innovation and advancing transplant capabilities are increasingly interconnected, jointly enhancing access and outcomes for patients with bone marrow failure.
Unmet Needs in the Bone Marrow Failure Treatment Space
Unmet needs in the treatment of bone marrow failure remain significant, largely because current therapies often address symptoms rather than underlying causes. Many patients depend on supportive care such as blood transfusions and growth factors, which can temporarily improve blood counts but do not restore healthy marrow function.
Curative options, like hematopoietic stem cell transplantation, are limited by donor availability, transplant-related complications, and the fact that many patients, particularly older adults, are not ideal candidates. As a result, a substantial proportion of individuals continue to face life-threatening complications, including infections, anemia, and bleeding, despite receiving standard care.
Another major gap lies in the limited availability of targeted or disease-modifying treatments. For many forms of bone marrow failure, such as idiopathic aplastic anemia or inherited marrow failure syndromes, the biological drivers remain only partially understood, slowing the development of precision therapies. Even in conditions where immunosuppressive therapy helps, responses can be incomplete or temporary, and relapse is common.
There is a pressing need for therapies that can reliably regenerate or protect hematopoietic stem cells, reduce dependence on transfusions, and improve long-term survival and quality of life. Advances in gene therapy, novel immunomodulators, and regenerative medicine hold promise, but translation into widely accessible, effective treatments is still at an early stage, leaving a clear and urgent unmet medical need.
Emerging Disease‑modifying Therapies
Pipeline innovation is increasingly mechanism‑driven, targeting immune dysregulation, telomere biology, apoptotic pathways, and macrophage function to move beyond purely supportive paradigms. Emerging bone marrow failure therapies such as CK0801 (Cellenkos), EXG-34217 (Elixirgen Therapeutics), sonrotoclax [BGB-11417; BeOne Medicines (SHA: 688235)], and bexmarilimab + SoC [(Faron Pharmaceuticals (LON: FARN)] reflect a new wave of mechanism-driven approaches targeting immune dysfunction, telomere biology, apoptotic pathways, and macrophage activity, signaling meaningful progress toward more effective, disease-modifying treatment options.
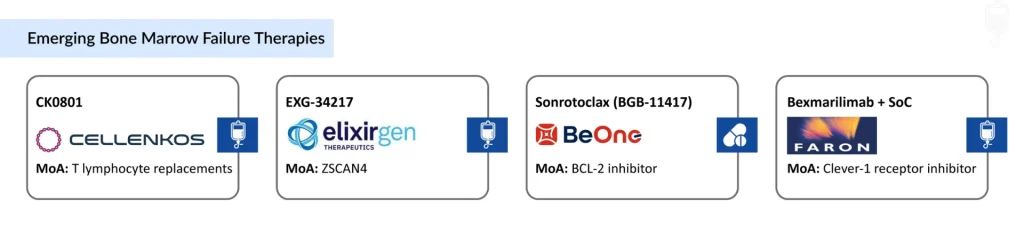
CK0801 is an allogeneic cord blood‑derived regulatory T‑cell product from Cellenkos designed to correct immune dysregulation in acquired aplastic anemia; it received FDA orphan drug designation in April 2025, highlighting its potential in a niche but high‑need segment. Elixirgen Therapeutics’ EXG‑34217 uses autologous CD34 hematopoietic stem cells transiently expressing the telomere‑regulator ZSCAN4 to induce telomere elongation and enhance genomic stability in telomere biology disorders with BMF, with early Phase III data reporting durable telomere lengthening in treated patients and no treatment‑related safety concerns over follow‑up of up to 24 months.
Sonrotoclax (BGB‑11417) from BeOne Medicines is an oral BCL‑2 inhibitor in development for myelodysplastic syndromes treatment, aiming to tilt the balance toward apoptosis in malignant clones while sparing normal progenitors. Bexmarilimab, a Clever‑1–targeting monoclonal antibody given with azacitidine, has demonstrated improved survival and evidence of hematopoietic recovery in higher‑risk MDS, including TP53‑mutated disease, in Phase III BEXMAB data presented at ASH 2025, signaling a potential shift in how clonal BMF is modulated immunologically.
The anticipated launch of these emerging bone marrow failure therapies are poised to transform the bone marrow failure market landscape in the coming years. As these cutting-edge bone marrow failure therapies continue to mature and gain regulatory approval, they are expected to reshape the bone marrow failure market landscape, offering new standards of care and unlocking opportunities for medical innovation and economic growth.
Future Outlook for Bone Marrow Failure Treatment
The bone marrow failure market across the 7MM reached approximately USD 1.4 billion in 2024, underscoring the sustained clinical burden and the ongoing dependence on foundational treatment approaches even as early innovation begins to shape future care pathways. In 2024, the US dominated the bone marrow failure market among the 7MM, capturing approximately 65% of the total 7MM market share.
According to Ramandeep Singh, Senior Consultant, Forecasting and Analytics at DelveInsight, the BMF market is limited by a few approved treatments and a high unmet need. However, pipeline innovation and rising awareness are expected to drive modest growth and improve future therapeutic options. Even though it is too soon to comment on the above-mentioned promising candidate to enter the market in the coming years, it is safe to assume that the future of this market is bright.
Agents such as CK0801, EXG‑34217, bexmarilimab plus azacitidine, and other late‑stage programs are anticipated to drive incremental growth in the BMF market and, more importantly, expand the therapeutic armamentarium beyond transplant and broad IST. As awareness, genetic testing, and risk‑adapted pathways improve, earlier diagnosis and more precise categorization of acquired versus inherited and clonal versus non‑clonal disease should enable better selection of candidates for HSCT, TPO‑R agonists, cellular therapies, and telomere‑targeted strategies.
Rising global healthcare spending and evolving reimbursement frameworks are expected to support modest but meaningful shifts from purely supportive care toward mechanism‑based, potentially disease‑modifying regimens, with the potential to transform long‑term outcomes in what remains a high‑mortality, high‑burden set of disorders.

Downloads
Article in PDF




