BYSANTI Approval Positions Vanda to Compete in the Antipsychotic Market
Mar 02, 2026
Summary
- The U.S. FDA approved Vanda Pharmaceuticals’ BYSANTI (milsaperidone) for the treatment of schizophrenia and manic or mixed episodes associated with bipolar I disorder.
- BYSANTI is built on the pharmacologic framework of FANAPT (iloperidone).
- Vanda’s stock surged nearly 40% after BYSANTI’s approval, reflecting renewed investor confidence.
- BYSANTI’s launch is expected by Q3 2026, contributing to portfolio diversification and revenue growth.
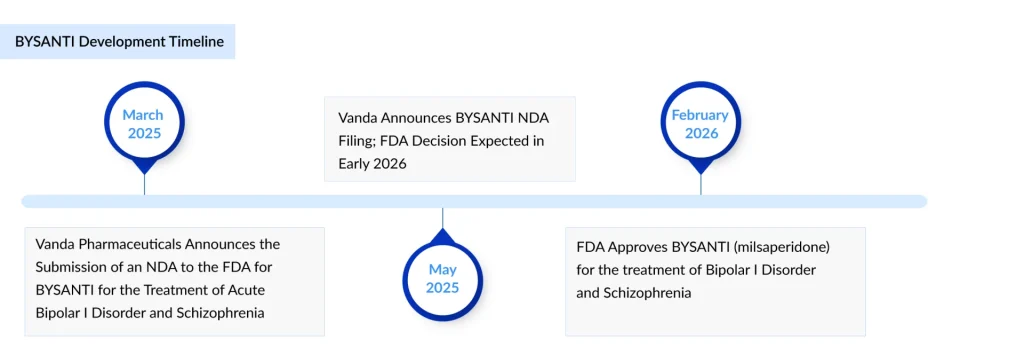
Vanda Pharmaceuticals’ atypical antipsychotic BYSANTI (milsaperidone) has won FDA approval for the treatment of schizophrenia and for manic or mixed episodes associated with bipolar I disorder, positioning the company back on offense in central nervous system (CNS) therapeutics after a high‑profile setback with its jet lag drug. Grounded in the pharmacology and extensive clinical heritage of Vanda’s existing antipsychotic FANAPT (iloperidone), BYSANTI is designed to offer prescribers a familiar mechanism in a new formulation that can be deployed from the front line of care.
The FDA has cleared BYSANTI as a frontline atypical antipsychotic for two major psychiatric indications: schizophrenia and manic or mixed episodes in patients with bipolar I disorder. These are overlapping but clinically distinct populations in which effective management of psychosis, mood instability, and functional impairment remains a major unmet need despite the availability of multiple antipsychotic classes.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Tackling Nausea and Vomiting Induced by Motion Sickness
- Vanda Broadens its Reach to Bipolar Disorder Treatment Market With Fanapt Approval
- Breaking Boundaries: Innovations and Updates in Schizophrenia Treatment
- FDA Grants Priority Review to BMS’ Luspatercept; Teva and MedinCell’s Risperidone FDA Approval; B...
- AUCATZYL Approved for R/R B-ALL; FDA Accepts NDA for Unicycive’s Oxylanthanum Carbonate; AstraZen...
According to Sadaf, Manager of Forecasting and Analytics at DelveInsight, BYSANTI’s approval allows clinicians to consider it early in the treatment algorithm, rather than relegating it to a niche or refractory-disease role. This positioning is important commercially and clinically because front‑line status shapes formulary negotiations, guideline inclusion, and real‑world prescribing patterns from the outset of launch.
Scientifically, BYSANTI is notable because it is based on iloperidone, the active moiety that forms the core of Vanda’s existing antipsychotic FANAPT. Milsaperidone, the active ingredient in BYSANTI, is an active metabolite of a compound that underlies FANAPT; once taken orally, BYSANTI rapidly interconverts to iloperidone in the body.
Iloperidone’s pharmacology is characterized by antagonism of dopamine D2 receptors, serotonin 5‑HT2A receptors, and alpha1‑adrenergic receptors, a receptor‑binding profile that is typical of many second‑generation, or “atypical,” antipsychotics. This multi‑receptor targeting underpins both antipsychotic efficacy and the nuanced side‑effect profiles that differentiate agents within the class.
By building BYSANTI around an already well‑understood pharmacologic scaffold, Vanda is not trying to reinvent antipsychotic treatment from scratch; instead, it is repackaging a known entity in a way that may offer practical and lifecycle advantages while preserving a familiar mechanism of action for prescribers.
Vanda’s development strategy for BYSANTI relied heavily on demonstrating its pharmacokinetic and clinical comparability to Fanapt. The company has shown that BYSANTI is bioequivalent to FANAPT, with pharmacokinetic profiles that follow similar trajectories over time when the drugs are administered. Such bioequivalence data suggest that systemic exposure to the active moiety is similar between the two products, supporting the inference that efficacy and safety should align closely.
Beyond direct bioequivalence trials, Vanda anchored its regulatory package in what it describes as the “well‑established knowledge of efficacy and safety” of FANAPT, encompassing more than 100,000 patient‑years of clinical and real‑world experience. This long‑term, large‑scale exposure record is critical in psychiatry, where chronic treatment is the rule rather than the exception and where rare but serious side effects must be well characterized.
In public statements around the approval, Vanda has emphasized that this heritage allows BYSANTI to be presented as a “reliable new treatment grounded in extensive clinical heritage,” highlighting continuity rather than novelty as a core part of its value proposition.
Schizophrenia and bipolar I disorder are both chronic, relapsing illnesses in which relapse prevention, symptom stabilization, and functional recovery require sustained pharmacologic management. An atypical antipsychotic with a known receptor profile and a large pre‑existing dataset can be attractive to clinicians who must often balance efficacy, tolerability, and patient preference over the long term.
BYSANTI’s shared indication set with FANAPT, schizophrenia, and manic or mixed episodes in bipolar disorder, means that many prescribers will conceptualize it within an already familiar clinical space. In practice, this can facilitate more rapid adoption if physicians perceive BYSANTI as offering comparable benefits with potential advantages in formulation, titration, or commercial access, even without a fundamentally new mechanism.
The timing of BYSANTI’s approval is strategically meaningful for Vanda. Earlier in the year, the FDA rejected the company’s attempt to expand its sedative Hetlioz into jet lag disorder, arguing that the submitted data did not adequately demonstrate efficacy in that setting. Vanda publicly disputed that conclusion, pointing out that the experimental models it used are widely accepted in circadian rhythm research as surrogates for eastward jet lag–related circadian misalignment.
That rejection was not an isolated event but part of a protracted dispute between the company and the agency over the jet lag indication. Hetlioz was first rejected in 2019, prompting Vanda to appeal and, after limited agency engagement, to sue the FDA in 2022. A federal judge in March 2024 ordered the agency either to grant Vanda a hearing or resolve the application; the FDA chose to resolve it, which led to another appeal by Vanda that it subsequently won in August 2025. Against that backdrop, a clean, positive decision on BYSANTI provides reputational and strategic relief, signaling that Vanda can still navigate complex CNS approvals despite past regulatory friction.
BYSANTI’s green light also comes on the heels of another key CNS win for Vanda: the FDA’s approval of NEREUS for motion sickness late last year, which was described as the first approval for this indication in more than four decades. That milestone, combined with BYSANTI and the ongoing commercial presence of Fanapt and Hetlioz in other indications, underscores Vanda’s ambition to be a diversified CNS company rather than a single‑asset player.
In this context, BYSANTI does more than add one more SKU to the portfolio. Paired with Nereus, it demonstrates that Vanda can deliver both incremental and category‑redefining assets across different parts of the CNS spectrum, from psychotic disorders and mood instability to acute vestibular symptoms like motion sickness.
Vanda has indicated that it expects to make BYSANTI available by the third quarter of the year following approval. The market responded positively to the announcement, with Vanda’s share price climbing nearly 40% between the close of trading on the day of the approval and the following Monday’s pre‑market session. Such a move reflects investor expectations that BYSANTI will contribute meaningfully to revenue and help de‑risk the company in light of its contentious regulatory history with Hetlioz.
Sadaf further said that BYSANTI enters a crowded atypical antipsychotic landscape, but does so with the advantage of a thoroughly characterized active moiety and a regulatory label that supports frontline use in two of the most burdensome psychiatric conditions. Clinicians who are already comfortable with FANAPT’s efficacy and tolerability profile may be particularly inclined to consider BYSANTI for appropriate patients if it offers practical advantages in dosing or access, even absent a radical mechanistic shift.
Taken together, BYSANTI’s approval, NEREUS’ recent success in motion sickness, and the long‑running but still active saga around Hetlioz’s jet lag application paint a picture of a company at a strategic inflection point. BYSANTI is not a moon‑shot radical innovation; instead, it is a carefully structured, evidence‑anchored evolution of a known drug, designed to consolidate Vanda’s position in the antipsychotic market while signalling to regulators, investors, and clinicians that the company can execute on complex CNS programs.
In an era when psychiatric drug development remains scientifically challenging and commercially risky, a product like BYSANTI, rooted in deep clinical heritage but packaged as a new, frontline‑ready option, may represent a pragmatic model for innovation: one that emphasizes reliability, regulatory tractability, and portfolio cohesion as much as it does mechanistic novelty.

Downloads
Article in PDF
Recent Articles
- How are Antipsychotics Transforming the Schizophrenia Treatment Space?
- Mitochondria destruction for Cancer Treatment; Atsena raises USD 55 Million financings; Novartis ...
- Business Cocktail
- Tackling Nausea and Vomiting Induced by Motion Sickness
- Hope on the Horizon: Brilaroxazine’s Promise for Schizophrenia Patients