Agitation in Alzheimer’s Disease Treatment Landscape: A New Dawn With AUVELITY’s Breakthrough Approval
May 11, 2026
Table of Contents
Summary
- In the United States alone, agitation in Alzheimer’s disease represented nearly 5 million diagnosed cases, highlighting a considerable yet often underappreciated burden among patients.
- Brexpiprazole (REXULTI) marked a key milestone as the first FDA-approved therapy specifically indicated for agitation in Alzheimer’s dementia, addressing a long-standing unmet need in behavioral symptom management.
- In late April 2026, Axsome Therapeutics reached a significant milestone when the FDA approved AUVELITY for treating agitation associated with dementia due to Alzheimer’s disease. This marked the first-ever approval of an oral, non-antipsychotic therapy for this condition.
- Emerging pipeline candidates such as IGC-AD1 (IGC Pharma), Masupirdine (SUVN-502) (Suven Life Sciences), EXV-801 ± Dextromethorphan (Exciva GmbH), COBENFY (KarXT; xanomeline–trospium chloride) (Bristol Myers Squibb/Karuna Therapeutics), and others are redefining the treatment landscape through diverse mechanisms of action.
- The market for agitation associated with Alzheimer’s disease across the 7MM exceeded USD 600 million in 2025, underscoring increasing clinical attention and commercial interest in managing this significant neuropsychiatric complication within the broader Alzheimer’s disease spectrum.
The “silent storm” of Alzheimer’s disease is no longer just about memory loss. Agitation, marked by restlessness, emotional distress, irritability, and verbal or physical aggression, is emerging as one of the most clinically challenging and therapeutically underserved manifestations of Alzheimer’s disease. With the landmark FDA approval of AUVELITY in April 2026, the treatment paradigm is shifting in a meaningful new direction.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Does coconut oil help prevent dementia?
- IMFINZI’s AEGEAN Phase III Trial Data for Treating Resectable NSCLC; Ipsen and Day One’s Exclusiv...
- Bristol Myers’ Opdivo combo Opdualag for Melanoma; Biogen’s Aduhelm; Marinus’ Ztalmy for CDKL-5 D...
- Quest Introduced Consumer-Initiated Blood Test for Alzheimer’s Disease Risk Assessment; DuP...
- Biochips in Healthcare: 8 Groundbreaking Applications Shaping the Industry
The Unspoken Burden: How Widespread Is Agitation in AD?
Agitation is far more prevalent than many realize. Up to 80% of individuals living with Alzheimer’s disease may experience agitation at some point during the progression of their illness. Nearly half of the patients with Alzheimer’s disease suffer from agitation. The number and severity worsen over time. According to DelveInsight’s analysis, approximately 5 million diagnosed cases of agitation in Alzheimer’s disease were recorded in the US alone, with the total across the seven major markets reaching around 11 million diagnosed cases in 2025.
These aren’t just numbers. Behind every statistic is a patient who paces in distress, a caregiver worn down by unpredictable outbursts, and a healthcare system struggling to respond effectively. Agitation significantly reduces quality of life, precipitates earlier institutionalization, and accelerates functional decline.
Current Agitation in Alzheimer’s Disease Treatment Landscape: Stuck in an Off-Label Rut
Despite the enormous disease burden, the treatment landscape for agitation in AD has historically been marked by improvisation rather than precision. Real-world prescribing data reveal that atypical antipsychotics are the most frequently prescribed agents (55.5% of prescriptions), followed by SSRIs (29.3%) and benzodiazepines (23.2%), all used off-label.
Drugs like risperidone, quetiapine, olanzapine, and haloperidol have been the workhorses of management, yet none were ever approved specifically for agitation in AD. Their use is shadowed by serious risks: sedation, falls, metabolic side effects, cardiovascular events, and, most alarmingly, increased mortality in elderly patients.
As DelveInsight’s analysis underscores, the continued reliance on off-label antipsychotics reflects a “structural gap” in care, where modest efficacy is frequently offset by clinically meaningful safety risks.
Non-pharmacological approaches, including structured routines, caregiver education, music therapy, and environmental modifications, remain foundational, but they are difficult to scale and cannot always address severe symptoms.
The First Approved Therapy: REXULTI Breaks the Ice
The first crack in the ceiling came in 2023, when brexpiprazole (REXULTI), co-developed by Otsuka Pharmaceutical and Lundbeck, became the first FDA-approved drug specifically for agitation associated with Alzheimer’s dementia. Operating through serotonin–dopamine activity modulation, partial agonism at 5-HT1A and D2 receptors, and antagonism at 5-HT2A receptors, it offered a more targeted mechanism compared to broadly acting antipsychotics. The approval was later extended to Japan in September 2024, signaling international regulatory recognition of this indication.
Despite this milestone, brexpiprazole’s reach is limited. With a US WAC of approximately USD 1,515 and an estimated 12-week treatment cost of ~USD 4,500 per patient, accessibility and reimbursement remain barriers. It paved the way, but the landscape needed more.
The Game Changer: FDA Approves AUVELITY for AD Agitation
In late April 2026, Axsome Therapeutics made history when the FDA approved AUVELITY (dextromethorphan hydrobromide/bupropion hydrochloride) for agitation associated with dementia due to Alzheimer’s disease, making it the first oral non-antipsychotic treatment ever approved for this indication.
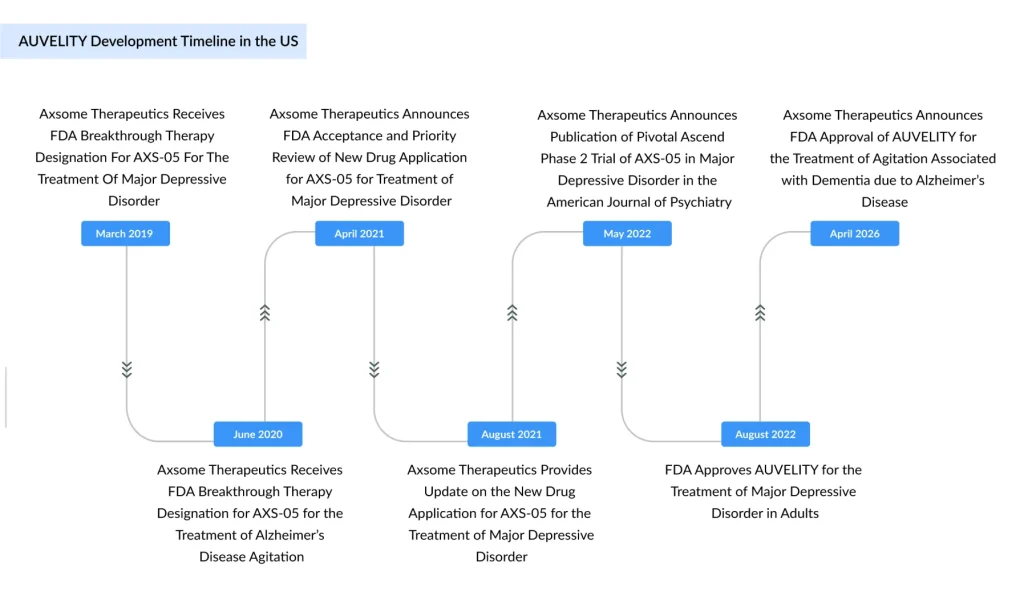
AUVELITY’s mechanism is what makes it uniquely compelling. It targets NMDA receptors and sigma-1 receptors, operating on glutamatergic and neuroplasticity pathways, an entirely different biological approach from conventional dopamine-based antipsychotics. This is not merely an incremental improvement; it represents a mechanistic breakthrough in how the neuroscience community is thinking about behavioral disturbances in AD.
The FDA approval of AUVELITY is backed by a robust, comprehensive clinical program anchored by two landmark Phase 3 studies, ADVANCE-1 and ACCORD-2. The significance of this approval is underscored by the prestigious regulatory designations it earned along the way. AUVELITY received both FDA Breakthrough Therapy Designation and Priority Review Designation for this indication, reserved for drugs addressing serious unmet medical needs with preliminary evidence of substantial improvement over existing therapies.
It is worth noting that AUVELITY was originally approved in 2022 for major depressive disorder (MDD) in adults, and this new indication dramatically expands its clinical and commercial footprint.
The Emerging Agitation in Alzheimer’s Disease Pipeline: What’s Coming Next?
The approval of AUVELITY is not an endpoint; it is a signal that the field is heating up. A pipeline of targeted agitation in alzheimer’s disease therapies is racing toward the finish line: IGC-AD1 (IGC Pharma), Masupirdine (SUVN-502) (Suven Life Sciences), EXV-801 ± Dextromethorphan (Exciva GmbH), COBENFY (KarXT; xanomeline–trospium chloride) (Bristol Myers Squibb/Karuna Therapeutics), and others.
Masupirdine is a selective 5-HT6 receptor antagonist, offering a non-dopaminergic pathway to symptom stabilization. Its global Phase III trial achieved 50% enrollment by November 2025, with topline data anticipated in Q1/Q2 2027. COBENFY, already gaining attention in schizophrenia, is now targeting agitation in AD through muscarinic receptor modulation, a cholinergic approach with potentially favorable cognitive benefits. IGC-AD1 uniquely combines melatonin and THC to address both neuroinflammatory and circadian rhythm-related contributors to agitation, with ~70% enrollment completed in its Phase II CALMA trial as of February 2026.

EXV-802, a Phase II/III neuroactive therapeutic candidate, is being evaluated both as a monotherapy and in combination with EXV-801 for its ability to modulate multiple central nervous system (CNS) signaling pathways implicated in agitation. This strategy reflects a broader shift toward multi-target or combination approaches designed to improve therapeutic efficacy while preserving functional tolerability in complex behavioral disorders. In November 2024, Exciva GmbH announced that its lead asset, EXV-802, was granted Fast Track Designation (FTD) by the US FDA for the treatment of agitation associated with Alzheimer’s disease.
Ramandeep Singh, neurology expert at DelveInsight, said that the emergence of novel, well-tolerated, and efficacious therapies presents a substantial market opportunity to address the growing unmet need in agitation in Alzheimer’s disease.
Agitation in Alzheimer’s Disease Market Opportunity: A Rapidly Growing Segment
The commercial opportunity in agitation associated with Alzheimer’s disease is rapidly expanding. In 2025, the agitation in Alzheimer’s disease market across the 7MM exceeded USD 600 million, with the United States accounting for nearly USD 500 million of that total. This growth is expected to accelerate significantly, with projections indicating the market will reach approximately USD 2.8 billion by 2036. This underscores one of the most significant unmet and commercially attractive opportunities in modern neurology.
At the same time, the treatment landscape is undergoing a meaningful transformation. What was once dominated by off-label therapies and limited clinical efficacy is now shifting toward a more structured, innovation-driven market characterized by approved, mechanism-based treatments and a robust mid-to-late-stage pipeline.
With increasing clinical and commercial momentum, the space is poised for substantial evolution, and by 2036, COBENFY is expected to emerge as the leading revenue-generating therapy across the 7MM for this indication, as per Singh.
For pharmaceutical companies, the message is clear: the agitation in the Alzheimer’s disease market is no longer a therapeutic backwater; it is one of the most strategically important frontiers in CNS drug development. With AUVELITY lighting the way, the race to own this space is officially on.

Downloads
Article in PDF
Recent Articles
- Biogen’s Aduhelm; FDA Approves Sanofi’s Enjaymo; NHS & Orchard Signs a Deal; Bri...
- Suneva Medical-Viveon Health merger update; Smith+Nephew acquires Engage Surgical; Abiomed’...
- A Deep Dive into the 17th Clinical Trials on Alzheimer’s Disease (CTAD) Conference
- Zenas BioPharma’s Obexelimab Delivers Positive Phase 3 Results in IgG4-RD; Sanofi Secures US Prio...
- Alzheimer’s Treatment: Latest Breakthroughs and Advancements in Therapeutic Development




