AI Integration in Radiopharmaceuticals: Transforming Precision Diagnostics and Targeted Therapy
Apr 01, 2026
Table of Contents
Radiopharmaceuticals are a cornerstone of precision medicine, enabling clinicians to visualize disease at the molecular level while simultaneously delivering targeted radionuclide therapy to tumors and other pathological tissues. Their dual diagnostic and therapeutic potential has positioned them as a transformative modality in modern healthcare. Now, the integration of artificial intelligence (AI) is reshaping this field, streamlining everything from target identification and ligand development to image interpretation, personalized dosimetry, and adaptive treatment planning, ultimately enhancing diagnostic accuracy and therapeutic safety.
At their core, radiopharmaceuticals are engineered by coupling a radioactive isotope with a biologically active molecule, enabling diseases to be visualized or treated through nuclear medicine modalities such as positron emission tomography (PET) and single-photon emission computed tomography (SPECT). These agents can either provide highly sensitive insights into molecular targets for diagnosis or deliver localized cytotoxic radiation to diseased tissues while sparing most healthy cells.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Cordis FDA Approval for MYNX CONTROL VENOUS Vascular Closure Device; Forma Medical’s Optimal Pla...
- IDEAYA and Servier Reports Successful Uveal Melanoma Trial; SynOx Therapeutics Highlights Positiv...
- 8 Key Applications of Radioligand Therapies in Healthcare
- Medical Radiation Detection Devices: Essential Tools in Healthcare and Diagnostics
- Diagnostic Precision: The Rise of Medical Imaging Technologies and Market Trends
Building on this foundation, radioligand therapies (RLTs) represent a highly targeted subclass of radiopharmaceuticals, in which a ligand binds selectively to tumor-specific receptors and carries a radioactive payload directly to cancer cells. This dual-component design allows for precise tumor targeting and controlled radiation delivery, forming the basis of many modern precision oncology approaches.
The convergence of these capabilities has driven the rise of theranostics, an integrated paradigm where matched diagnostic and therapeutic agents target the same biological pathway. In practice, imaging is first used to confirm the presence of a specific molecular target, after which a corresponding therapeutic radioligand is deployed to treat the disease. This approach is already reshaping treatment strategies, particularly in oncology areas such as neuroendocrine tumors and prostate cancer, and is central to the growing clinical adoption of radioligand-based precision medicine.
However, despite these advancements, the development and clinical implementation of radiopharmaceuticals remain complex, time-consuming, and resource-intensive, requiring the navigation of intricate chemical, biological, and regulatory challenges. AI and machine learning are increasingly addressing these barriers by leveraging large, multimodal datasets to accelerate discovery, refine molecular design, automate imaging and dosimetry workflows, and enable truly personalized treatment strategies at scale.
The Role of Radiopharmaceuticals in Modern Medicine
Radiopharmaceuticals enable non-invasive visualization of gene expression, receptor status, metabolism, perfusion, and other functional processes that are invisible to conventional anatomical imaging. Clinically, they are used across oncology, cardiology, neurology, and inflammatory diseases for early diagnosis, staging, response assessment, and long-term follow-up.
Therapeutic radiopharmaceuticals, such as lutetium-177–labeled peptides and small molecules, deliver targeted radionuclide therapy that induces irreparable DNA damage in tumor cells while better sparing normal tissues compared with external beam radiotherapy. FDA approvals of radiotheranostic pairs like [177Lu]Lu-DOTA-TATE/[68Ga]Ga-DOTA-TATE and [177Lu]Lu-PSMA-617/[68Ga]Ga-PSMA-11 have demonstrated the power of using a diagnostic agent to stratify patients, predict response, and guide individualized therapy dosing.
As indications expand into neurodegenerative, cardiovascular, and inflammatory conditions, the need for more diverse, better-optimized radiopharmaceuticals and more efficient development pipelines continues to grow.
How AI Is Transforming Radiopharmaceutical Development
AI is accelerating radiopharmaceutical development by enabling faster target identification, optimizing radiotracer design, and improving predictive modeling of biodistribution and toxicity. Advanced algorithms can analyze imaging and molecular data to streamline clinical trial design and patient selection. This ultimately reduces development timelines and enhances precision in targeted diagnostics and therapies.
Accelerating Target Identification and Drug Discovery
AI models are being applied to high-dimensional omics data, biological networks, and imaging-derived biomarkers to identify novel molecular targets suitable for radiotheranostic development. Techniques such as graph neural networks (GNNs), transformers, and network-based inference can integrate genomic, transcriptomic, proteomic, and protein–protein interaction data to prioritize targets with favorable expression patterns, accessibility, and disease relevance.
In parallel, AI-driven virtual screening and in silico drug design significantly reduce the time and cost required to discover ligands with high affinity, specificity, and suitable pharmacokinetics for radiolabeling. Deep learning models for drug–target affinity prediction, molecular docking surrogates, and generative models can propose, rank, and iteratively optimize candidate scaffolds before any wet-lab synthesis, shortening the discovery cycle from years to months in some small-molecule programs.
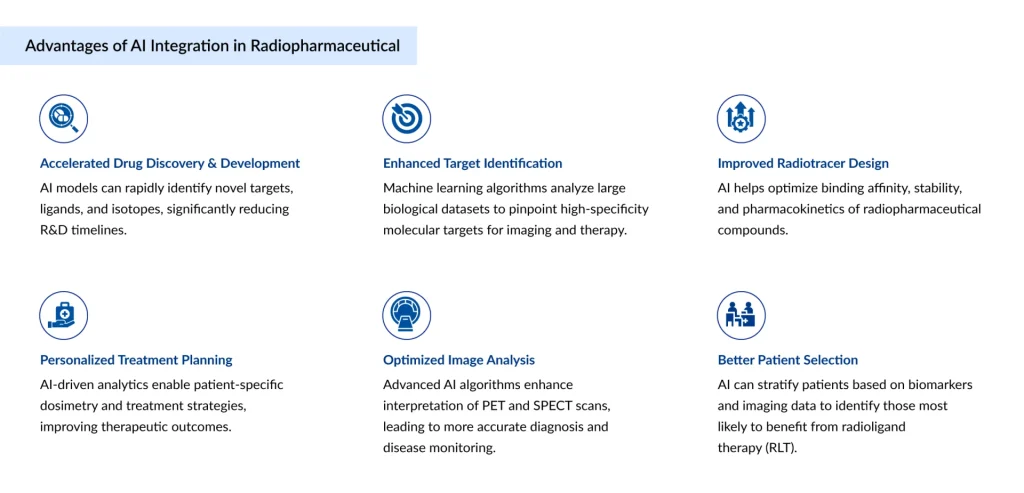
AI-Driven Radiochemistry and Molecule Design
The same AI methodologies transforming general drug discovery are being tailored to address radiopharmaceutical-specific constraints, such as radionuclide compatibility, chelator geometry, and radiation stability. Generative models can design ligands that integrate both binding and metal-coordination features, optimizing for target affinity and radiolabeling efficiency simultaneously as a multi-objective problem.
Reinforcement learning has been proposed to fine-tune radiochemical synthesis conditions, including temperature, pH, precursor amount, and reaction time, to maximize labeling yield and minimize by-products in short-lived radionuclide workflows. Machine learning models that link chelator structure to physicochemical and in vivo stability properties provide rational guidance for designing more robust radiometal conjugates.
Enhancing Imaging Analysis and Quantification
AI is driving major advances in PET/SPECT image reconstruction, segmentation, and quantitative analysis, all critical for both diagnostic accuracy and therapy planning. Deep learning–based reconstruction can denoise low-count or low-dose acquisitions, improve spatial resolution, and enable shorter scan times without compromising quantitative fidelity.
Convolutional neural networks and hybrid architectures provide automated, highly reproducible segmentation of tumors and organs-at-risk, facilitating the extraction of radiomics features and standardized uptake values (SUVs) for large patient cohorts. AI-based partial-volume effect correction and attenuation/scatter correction further improve quantitative accuracy, which is essential for reliable dosimetry in radiopharmaceutical therapy.
Personalized Dosimetry and Treatment Planning
Routine patient-specific dosimetry is increasingly recognized as essential for safe, effective radiopharmaceutical therapy, but is often perceived as too complex and time-consuming for widespread clinical adoption. AI is emerging as a key enabler of simplified, patient-centered dosimetry workflows by learning dose–response relationships and time–activity curves from limited imaging and clinical data.
Deep learning models can estimate voxel-level or organ-level absorbed doses from reduced imaging protocols (fewer time points or simplified views), significantly lowering the acquisition burden while maintaining acceptable accuracy. AI-enabled segmentation and registration pipelines also automate critical steps in dosimetry, such as organ delineation, lesion tracking, and image alignment across time points, supporting individualized treatment planning and adaptive retreatment strategies.
Improving Clinical Trial Design and Patient Selection
AI applied to multimodal datasets, combining PET/SPECT imaging, genomics, pathology, and clinical variables, can refine eligibility criteria, enrich trials for likely responders, and identify early imaging biomarkers of response or toxicity in radiopharmaceutical studies. Radiomics and deep learning signatures derived from baseline and interim scans show promise for predicting outcomes in theranostic trials, which can inform dose escalation, retreatment decisions, and combination strategies.
Simulation-based trial design using synthetic patient cohorts generated from historical imaging and dosimetry data may further optimize sample sizes, endpoint selection, and imaging schedules, reducing cost and accelerating regulatory approval for novel radiopharmaceuticals.
Challenges in AI Adoption
AI adoption in radiopharmaceuticals is constrained first by data issues: models need large, diverse, and well-annotated datasets, yet clinical information is fragmented across centers, vendors, and modalities, with heterogeneous imaging protocols and dosimetry workflows. Privacy regulations and relatively small patient cohorts in many theranostic indications further limit central data pooling, making it difficult to train and validate robust, generalizable models. Emerging approaches like multicenter data standardization and federated learning, which allow model training across institutions without moving raw data, are promising but still early in real-world nuclear medicine deployments.
A second barrier is trust: many high-performing AI models operate as black boxes, which clashes with the need for transparency, interpretability, and clear uncertainty estimates in dose calculations, image interpretation, and treatment decisions. Regulators and clinicians expect rigorous external validation across scanners, centers, and populations, along with predictable behavior and explicit performance benchmarks before integrating AI outputs into clinical decision-making. Ethical and legal concerns, around bias, equity, accountability, informed consent for secondary data use, and explainability, also slow approvals and uptake if not proactively addressed through strong governance and oversight frameworks.
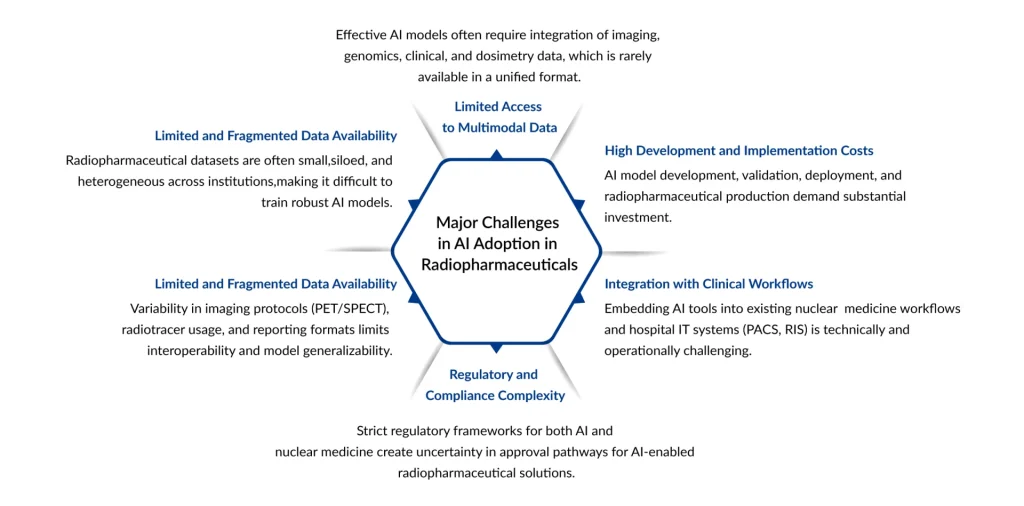
Finally, workflow integration and user acceptance are critical practical challenges: nuclear medicine services are under significant time and staffing pressure, so any AI tool that adds clicks, complexity, or uncertainty rather than streamlining tasks will struggle to gain traction. Lack of unified AI platforms, limited interoperability with PACS, oncology information systems, and radiopharmacy software, and insufficient training for clinicians and physicists can all hinder real-world implementation and post-deployment monitoring. Meaningful involvement of end users in design, intuitive interfaces, and clearly actionable outputs are therefore as important as algorithmic accuracy for sustainable AI adoption in radiopharmaceutical practice.
The Future of AI-Enabled Radiopharmaceuticals
Looking ahead, AI is poised to enable closed-loop radiotheranostic ecosystems in which target discovery, ligand design, imaging, dosimetry, and outcome monitoring continuously inform each other. Dedicated databases and knowledge graphs for radiopharmaceuticals, such as emerging curated resources cataloguing agents, targets, and clinical data, will further support AI-driven discovery and repurposing.
The global radiopharmaceuticals market is projected to grow significantly, rising from USD 10.7 billion in 2025 to USD 24.1 billion by 2034, indicating robust and sustained expansion at a CAGR of 9.5%. This growth is largely driven by the increasing burden of chronic diseases, including cancer, cardiovascular disorders, and neurological conditions, which is fueling demand for advanced diagnostic and therapeutic solutions, as per DelveInsight.
Additionally, the active participation of key companies such as Lantheus, Cardinal Health, GE Healthcare, Curium Pharma, Pharmalogic Holdings Corp., Novartis, Cambridge Isotope Laboratories, Inc., Sotera Health LLC, Bracco Imaging SpA, Jubilant Pharma Limited, NECSA Ltd., IBA Radiopharma Solutions, Bayer AG, Norgine, NorthStar Medical Radioisotopes, LLC, Eckert & Ziegler, Telix Pharmaceuticals Limited, ITM Isotope Technologies Munich SE, Mallinckrodt PLC, ROTOP Pharmaka GmbH, and others will further propel the market growth.
In parallel, theranostics and AI are converging to create highly adaptive, patient-specific treatment pathways in oncology and beyond, where diagnostic scans not only identify disease but also drive automated dosimetry, therapy selection, and longitudinal response prediction. As regulatory and ethical frameworks mature, and as more prospective evidence accumulates, AI-enabled radiopharmaceuticals are likely to become a cornerstone of precision diagnostics and targeted therapy, reshaping how complex diseases are detected, characterized, and treated across the patient journey.

Downloads
Article in PDF
Recent Articles
- 8 Key Applications of Radioligand Therapies in Healthcare
- Medical Radiation Detection Devices: Essential Tools in Healthcare and Diagnostics
- Evolving Landscape of Anti-GPC3 Targeted Therapies: ADCs, CAR-T Cells, and Beyond
- Cordis FDA Approval for MYNX CONTROL VENOUS Vascular Closure Device; Forma Medical’s Optimal Pla...
- IDEAYA and Servier Reports Successful Uveal Melanoma Trial; SynOx Therapeutics Highlights Positiv...