HER2-NSCLC Treatment Landscape: From Late-Line Breakthroughs to Frontline Transformation
May 15, 2026
Table of Contents
Summary
- NSCLC accounts for nearly 80–85% of all lung cancer cases. Among these patients, HER2 mutations are identified in roughly 1–4% of cases.
- ENHERTU became the first FDA-approved therapy for unresectable/metastatic HER2 (ERBB2)-mutant NSCLC in 2022; however, along with recently approved HERNEXEOS and HYRNUO, treatment remains largely limited to the second-line setting, highlighting a persistent unmet need in first-line HER2-mutant NSCLC.
- Potential drugs in the pipeline include Pyrotinib (Jiangsu HengRui Medicine), FWD1509 (Forward Pharmaceuticals), IAM1363 (Iambic Therapeutics), EO-1001 (Edisononcology), and others in different stages of clinical development.
HER2, encoded by the ERBB2, is a transmembrane receptor tyrosine kinase that regulates cell proliferation and survival. In NSCLC, HER2 alterations define a distinct molecular subset with unique biology and emerging therapeutic relevance. HER2 alterations in NSCLC comprise three biologically and clinically distinct subsets: Mutations, amplification, and overexpression. HER2 mutations, most commonly Exon 20 insertions, occur in approximately 1–4% of patients and represent true oncogenic drivers with clear sensitivity to targeted therapies. Beyond this, HER2 amplification is observed in ~4% of cases and reflects increased ERBB2 gene copy number, although its role as a predictive biomarker remains less consistent. In contrast, HER2 overexpression, identified in ~16% of NSCLC cases based on protein expression, represents the largest subgroup but is often not directly linked to underlying genomic alterations, contributing to variable therapeutic outcomes.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Lilly’s Morphic Acquisition; IDEAYA’s IDE397 Positive Phase II Trial Result; XPOVIO (seline...
- Potential of EGFR Inhibitors: A Promising Avenue in Cancer Treatment
- AstraZeneca’s Imfinzi for Biliary Tract Cancer; FDA Clears Boehringer’s Spesolimab; Novo Nordisk ...
- ENHERTU: Another Triumph to Celebrate for AstraZeneca and Daiichi Sankyo
- The Next Chapter in NSCLC Treatment Space: Recent Discoveries and Innovations
Despite early recognition of HER2 as a potential oncogenic driver, therapeutic targeting in NSCLC remained challenging for years, particularly outside the mutation-defined subset. Initial approaches failed to demonstrate meaningful clinical benefit, leaving patients dependent on non-specific regimens. As a result, platinum-based chemotherapy combined with immune checkpoint inhibitors became the Standard of Care (SoC), delivering modest outcomes with Objective Response Rates (ORR) of around 43% and a median Progression-free Survival (PFS) of approximately 6 months. These outcomes underscore a clear gap between HER2-altered NSCLC and more mature biomarker-driven segments.
First Breakthrough: ADCs Open the Door, TKIs Drive the Next Wave
A turning point came in 2022 with the approval of trastuzumab deruxtecan (T-DXd, sold as ENHERTU), developed by AstraZeneca and Daiichi Sankyo, the first HER2-targeted ADCs to demonstrate significant efficacy in this setting. Approved for patients with unresectable or metastatic HER2-mutant NSCLC who have received prior systemic therapy, the drug is positioned in the second-line and later setting. Supported by results from the Phase II DESTINY-Lung02 trial, the therapy achieved a cORR of 57.7% in previously treated patients, along with a mDoR of 8.7 months. These results marked a clear improvement over historical outcomes and, importantly, established proof-of-concept for HER2-directed therapy in NSCLC.
However, while this milestone validated HER2 as a druggable target, it did not fully resolve the treatment challenge. The reliance on accelerated approval based on response rates, coupled with considerations around durability and safety, highlighted that further innovation was still required. In this sense, ADCs such as trastuzumab deruxtecan (T-DXd, ENHERTU) laid the foundation but not the final answer for HER2-targeted treatment in NSCLC.
The landscape continued to evolve with the emergence of next-generation HER2 Tyrosine Kinase Inhibitors (TKIs). In August 2025, Zongertinib (HERNEXEOS), developed by Boehringer Ingelheim, was initially approved for patients with unresectable or metastatic HER2-mutant NSCLC who had progressed on prior systemic therapy. In the Phase I/II Beamion LUNG-1 trial, zongertinib demonstrated strong clinical activity, achieving an ORR of 75% in previously treated patients, with more than half of responses lasting at least 6 months. Importantly, it also retained activity in patients who had received prior HER2-targeted ADCs, highlighting its potential in resistant disease settings.
The landscape continued to evolve with the approval of Sevabertinib (HYRNUO) in November 2025, marking the entry of selective HER2 TKIs into this space. Indicated for patients with unresectable or metastatic HER2-mutant NSCLC who have received prior systemic therapy, sevabertinib demonstrated strong efficacy in the Phase II SOHO-01 trial. In HER2 therapy naïve patients, it achieved a confirmed ORR of 71% with a median duration of response of 9.2 months. Notably, it also retained activity in patients previously treated with HER2-directed ADCs, with an ORR of 38%, underscoring its relevance in more resistant disease settings.
| Comparative Overview of HER2-targeted Therapies in 2L/2L+ NSCLC | ||||
| Drug | ROA and Dosing Frequency | Pivotal Study Data (Second-line and Above Segment) | The US Annual Price (in USD) | First Approval (The US) |
| Trastuzumab deruxtecan (ENHERTU) | 5.4 mg/kg IV infusion every 3 weeks | DESTINY-Lung02 (NCT04644237): cORR: 50%; mPFS: 9.9 monthsDESTINY-Lung01 (NCT03505710): cORR: 52.9%; mPFS: 8.2 months | ~165,000 | 2022 |
| Zongertinib (HERNEXEOS) | <90 kg: 120 mg orally once daily ≥90 kg: 180 mg orally once daily | Beamion LUNG-1 (NCT04886804): ORR: 75%; CR: 6%; PR: 69%: PFS: 12.4 months | ~254,000 | 2025 |
| Sevabertinib (HYRNUO) | 20 mg orally twice daily (with food) | SOHO-01 (NCT05099172): ORR: 71%; CR: 2.9%; PR: 69%; PFS: 8.3 months | ~200,000–215,000 | 2025 |
| cORR = confirmed objective response rate; ORR = objective response rate; CR = complete response; PR = partial response; mDoR = median duration of response; mPFS: median progression free survivalAssumptions for priceThe cost of Trastuzumab deruxtecan was estimated based on a price of USD 2,365.37 per 100 mg vial; assuming an average patient weight of 80 kg and a recommended dose of 5.4 mg/kg, a total dose of 432 mg per 21-day cycle requires approximately five vials per cycle, and with a mPFS of 9.9 months, this forms the basis for the overall treatment cost estimation.The list price for Sevabertinib (HYRNUO) is around USD 24,000 per month, compared to USD 21,172 for Zongertinib (HERNEXEOS) for one month of treatment. Zongertinib demonstrated a slightly higher mPFS (12.4 months) compared to the reported 8.3 months for Sevabertinib in a similar anti-HER2-naive, pre-treated population in previously treated, HER2-targeted-naive populations. | ||||
Oral HER2 TKIs offer several practical and clinical advantages over ADCs
- Convenience and accessibility: Oral administration avoids IV infusions, reducing hospital visits and improving patient compliance.
- Continuous target inhibition: Daily dosing enables sustained HER2 blockade, which may enhance disease control.
- Safety and tolerability: Generally lower risk of ADC-related toxicities such as interstitial lung disease and infusion-related reactions.
- Sequencing flexibility: Can be used before or after ADCs, including in patients progressing on prior HER2-directed therapies.
Unlocking the Frontline: Zongertinib Redefines the Treatment Paradigm
Building on this momentum, zongertinib represents the next phase of evolution in HER2-mutant NSCLC, transitioning from a post-line option to a potential earlier-line therapy. Notably, efficacy extended beyond previously treated patients, with treatment-naive cohorts demonstrating an ORR of 76% and durable responses in the majority of patients. This step-up in activity suggests that earlier HER2 pathway inhibition may translate into greater clinical benefit.
These data supported the February 2026 label expansion, removing the prior treatment requirement and enabling use in earlier-line settings, effectively accelerating its shift from salvage therapy toward frontline positioning. This rapid progression within a short timeframe marks a significant inflection point in the treatment paradigm.
“A 71% response rate is unprecedented in this cancer subtype, and not only is the data strong in showing that this treatment works, but zongertinib has the added convenience of being a once-daily oral therapy. When you also consider the improved safety profile compared to less selective inhibitors, this suggests a promising approach for patients in need of new treatments. That’s exciting because just a few years ago these patients had no effective targeted therapies.”
More broadly, it reflects a structural shift in HER2 NSCLC, where targeted therapies are no longer reserved for later lines but are increasingly moving upfront to maximize patient outcomes. The stepwise improvement in response rates from ~43% with chemo-immunotherapy to ~57.7% with ADCs and up to ~75–76% with TKIs further illustrates how successive waves of innovation are redefining expectations in this segment.
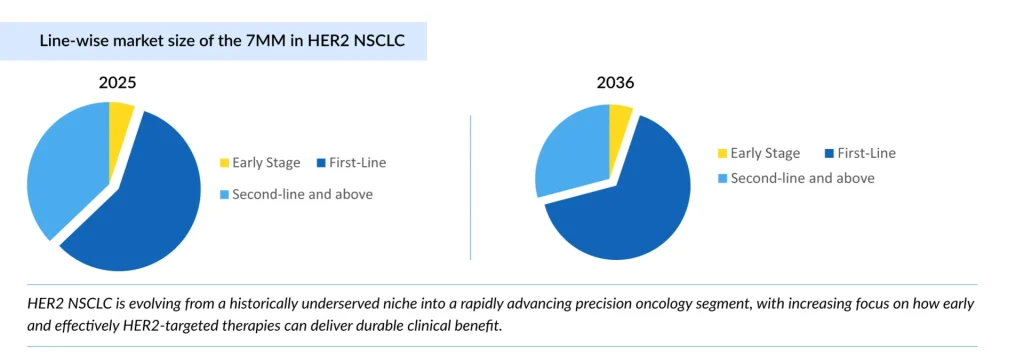
Looking ahead, the HER2 NSCLC treatment landscape is expected to evolve rapidly, with trastuzumab deruxtecan (ENHERTU) from AstraZeneca and Daiichi Sankyo expanding into earlier lines by 2027. Sevabertinib from Boehringer Ingelheim is expected to follow by 2028, while ENHERTU is further being explored in HER2 overexpression with a longer-term expansion timeline toward 2031. This progression highlights a shift toward a competitive, multi-asset, and earlier-line treatment paradigm in HER2 NSCLC.
In 2025, the HER2 NSCLC market across the seven major markets (the United States, Germany, France, Italy, Spain, the United Kingdom, and Japan) is estimated at nearly USD 1 billion, with growth primarily driven by the expanding first-line segment, reflecting a clear shift toward earlier use of targeted therapies. The market is projected to expand at a CAGR of 16.3% between 2026 and 2036 in the seven major markets.
What’s Next: Competitive Intensity and Pipeline Momentum?
Looking ahead, the HER2 NSCLC landscape is poised for continued transformation across first-line, later-line, and emerging adjuvant settings, reflecting a shift toward a multi-line treatment ecosystem. Ongoing Phase III trials, including Beamion LUNG-2 (Boehringer Ingelheim), are expected to provide confirmatory evidence and further define the positioning of HER2 TKIs within this continuum.

At the same time, combination strategies are emerging as a key area of innovation, particularly in earlier lines. Notably, BL-M07D1 in combination with Pembrolizumab (SystImmune) is being evaluated in HER2-overexpressing NSCLC, alongside broader chemo-immunotherapy combinations in frontline settings, reflecting efforts to enhance depth and durability of response.
In parallel, next-generation HER2 NSCLC agents such as sevabertinib (Bayer) and trastuzumab rezetecan (Hengrui Pharma/Glenmark Pharmaceuticals) are being developed across multiple lines of therapy, further intensifying competition and expanding treatment options.
As this space matures, the landscape is expected to evolve from a single-asset, later-line paradigm to a multi-asset, multi-line ecosystem, where differentiation will increasingly depend on durability, safety, and the ability to integrate into combination and sequencing strategies across the treatment continuum.
Commercial Opportunity: Expansion Beyond HER2-mutant NSCLC
HER2 NSCLC is transitioning from a historically underserved niche to a rapidly evolving precision oncology segment. The key question is no longer whether HER2 can be effectively targeted but how early and how well these therapies can be integrated to deliver meaningful and lasting benefit for patients.
In NSCLC, HER2 mutation is already a validated target, but HER2 overexpression and HER2 amplification remain much less clearly tapped commercially and clinically, so there is still room for key players to build a differentiated strategy. HER2 overexpression appears more common than HER2 mutation, so the addressable population could be larger if a treatment proves effective. HER2 amplification occurs in a smaller subset, but it is still clinically relevant and may represent a separate biomarker-defined opportunity.

Downloads
Article in PDF
Recent Articles
- ENHERTU: Another Triumph to Celebrate for AstraZeneca and Daiichi Sankyo
- CereVasc’s eShunt System Study; FDA Approves NGS-Based CDx for Trastuzumab Deruxtecan; Nanopath S...
- Bristol Myers’ Opdivo combo Opdualag for Melanoma; Biogen’s Aduhelm; Marinus’ Ztalmy for CDKL-5 D...
- Tumor‑Infiltrating Lymphocyte (TIL) Therapies: Redefining Cell Therapy for Solid Tumors
- FDA Approves Lexicon’s INPEFA (Sotagliflozin); PTC Therapeutics’s Vatiquinone MOVE-FA Registratio...