EGFR Market Summary
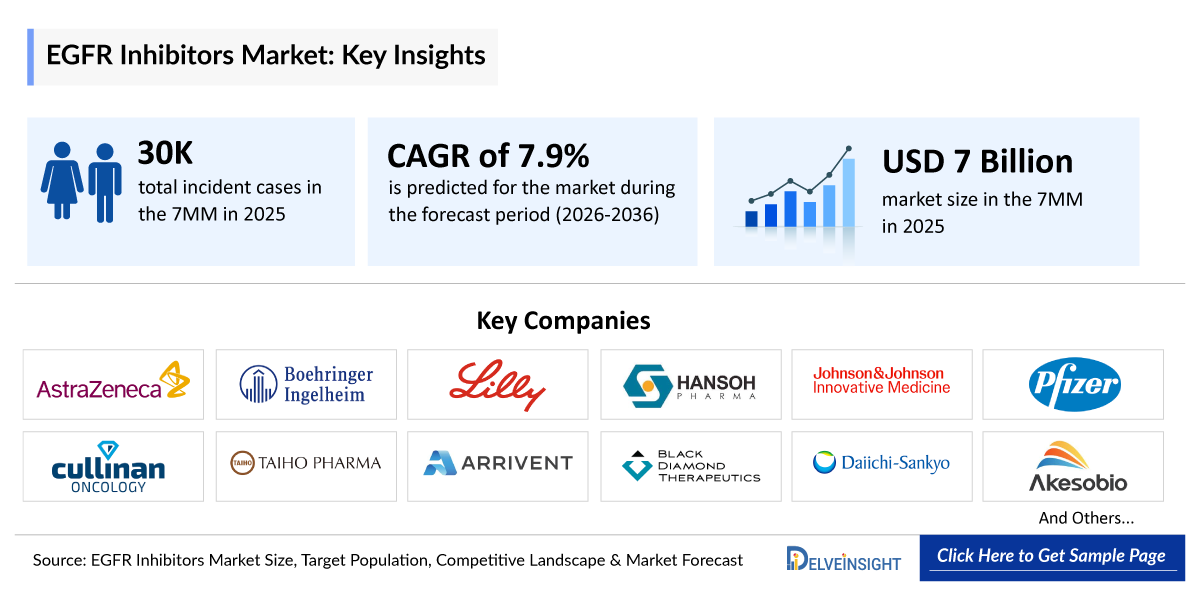
- According to DelveInsight’s analysis, EGFR Inhibitors market size was found to be ~USD 350,000 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
Epidermal Growth Factor Receptor (EGFR) Inhibitors Insights and Trends
- Germany shows consistent growth in NSCLC revenues, reflecting steady adoption of EGFR-targeted therapies and increasing treatment penetration. HNC revenues accelerate significantly after the late 2020s, suggesting entry of new targeted or biologic therapies that drive market expansion. Meanwhile, CRC revenues grow modestly, highlighting a stable but limited-growth market due to the presence of established treatment options.
- Across the seven major markets, NSCLC remains the dominant revenue-generating indication, driven by widespread adoption of EGFR-targeted therapies. HNC represents the fastest-growing segment, reflecting pipeline innovation and increasing treatment uptake, particularly in the US and Europe. Meanwhile, CRC demonstrates comparatively modest growth, highlighting a mature therapeutic landscape with fewer transformative treatment developments.
- The United States continues to be the largest market for EGFR-targeted therapies, with Non-Small Cell Lung Cancer (NSCLC) accounting for the majority of the revenue share during the forecast period. NSCLC revenues rise significantly until the early 2030s and then experience a slight decline toward 2036, reflecting market maturity and increasing competition from emerging treatments. Meanwhile, Head and Neck Cancer (HNC) records the fastest growth, expanding multiple times over the forecast timeline, which highlights growing therapeutic innovation and greater treatment adoption in this indication. In comparison, Colorectal Cancer (CRC) shows gradual and consistent growth, suggesting a relatively mature and stable treatment environment.
- Japan represents the second-largest EGFR therapy market after the United States, particularly driven by NSCLC, which maintains strong and stable growth throughout the forecast period. HNC revenues increase steadily, especially after 2030, reflecting gradual adoption of emerging therapies. CRC revenues show limited expansion, indicating a stable and mature market with incremental growth.
- In the 7MM, NSCLC is the least contributor. However, the incidence continues to rise due to several factors, including persistent tobacco smoking, increasing exposure to air pollution and occupational carcinogens and second-hand smoke.
- EGFR is also highly expressed in colorectal cancer (CRC), where it contributes to tumor cell growth and survival through activation of downstream signaling pathways such as RAS/RAF/MEK/ERK. CRC stands as the second-largest contributor, with the 7MM having 120,000 cases EGFR-expressing cases in 2025.
Epidermal Growth Factor Receptor (EGFR) Inhibitors Market Size and Forecast
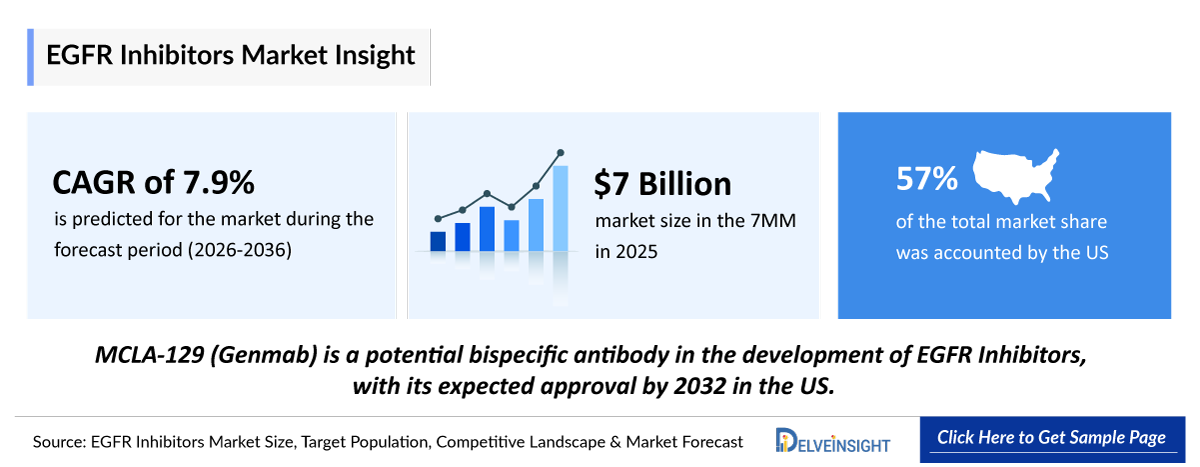
- 2025 EGFR Inhibitor Market Size: ~USD 7,000 million
- 2036 Projected EGFR Inhibitor Market Size: ~USD 16,000 million
- EGFR NSCLC Growth Rate (2026–2036): 7.9% CAGR
DelveInsight's ‘Epidermal Growth Factor Receptor (EGFR) Inhibitor – Market Size, Target Population, Competitive Landscape & Market Forecast – 2036’ report delivers an in-depth understanding of the EGFR inhibitors, historical and forecasted epidemiology, as well as the EGFR inhibitors market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Epidermal Growth Factor Receptor (EGFR) inhibitors market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, EGFR inhibitors patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in EGFR inhibitors and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
Scope of the EGFR Inhibitor Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Geographies Covered |
|
|
EGFR Inhibitor Market CAGR (Study period/Forecast period) |
7.9% (2026-2036) |
|
EGFR Inhibitor Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
EGFR Inhibitor Companies |
|
|
EGFR Inhibitor Therapies |
|
|
EGFR Inhibitor Market |
Segmented by
|
|
Analysis |
|
Epidermal Growth Factor Receptor (EGFR) Inhibitor Understanding and Treatment Algorithm
Epidermal Growth Factor Receptor (EGFR) Inhibitor Overview and Diagnosis
The Epidermal Growth Factor Receptor (EGFR) is a 170 kDa transmembrane glycoprotein and a key member of the ErbB receptor family, which also includes HER2, HER3, and HER4. Structurally, EGFR comprises an extracellular ligand-binding domain, a hydrophobic transmembrane region, and an intracellular tyrosine kinase domain that regulate key signaling pathways controlling cell proliferation, differentiation, migration, and survival.
Aberrant activation of EGFR, through gene mutations, amplification, or overexpression, is a major oncogenic driver in several cancers, particularly Non-small Cell Lung Cancer, with most mutations occurring in exons 18–21 of the EGFR gene. EGFR activation is initiated when ligands such as Epidermal Growth Factor, Transforming Growth Factor Alpha, amphiregulin, epiregulin, betacellulin, or HB-EGF bind to its extracellular domain, triggering receptor dimerization and autophosphorylation of intracellular tyrosine residues. These phosphorylated sites recruit adaptor proteins including Shc and GRB2, which subsequently activate downstream signaling cascades such as the MAPK signaling pathway, PI3K signaling pathway, PLC?–PKC signaling, and STAT pathways.
Collectively, these pathways regulate gene transcription and cellular responses; however, EGFR overactivation in tumors drives uncontrolled proliferation, angiogenesis, metastasis, and resistance to apoptosis, making it a critical therapeutic target in oncology.
Top-3 target indications in 7MM (EGFR-expressing incident cases) are NSCLC, CRC and Head and Neck cancer.
In Colorectal Cancer (CRC), activating EGFR mutations are uncommon; therefore, molecular diagnostics primarily focus on identifying downstream pathway alterations. Clinical guidelines recommend comprehensive genotyping for RAS (KRAS and NRAS) and BRAF mutations prior to initiating anti-EGFR therapy. These biomarkers are typically assessed using allele-specific PCR or NGS panels on FFPE tumor tissue, which enable simultaneous detection of multiple genomic alterations. Increasingly, circulating tumor DNA analysis through liquid biopsy is also used to identify actionable mutations and monitor resistance non-invasively.
In Head and Neck Cancer, EGFR mutations are relatively rare (=10%), whereas EGFR overexpression occurs in the majority of tumors. Molecular assessment is primarily conducted on tumor tissue using PCR-based assays, such as the Therascreen EGFR RGQ PCR Kit, which detects mutations in exons 18–21 with high sensitivity. Additional techniques, including high-resolution melting analysis, Sanger sequencing, and NGS panels, may be employed for confirmatory testing and broader genomic profiling, enabling detection of rare EGFR variants and other clinically relevant alterations.
Further details are provided in the report.
Current Epidermal Growth Factor Receptor (EGFR) Inhibitor Treatment Landscape
In Non-small Cell Lung Cancer (NSCLC), EGFR-targeted therapy primarily involves multiple generations of EGFR tyrosine kinase inhibitors (TKIs) that inhibit aberrant EGFR signaling. First-generation agents such as Gefitinib and Erlotinib reversibly inhibit the EGFR tyrosine kinase domain, while second-generation inhibitors including Afatinib and Dacomitinib provide irreversible inhibition but may be limited by toxicity and the emergence of the T790M resistance mutation. Third-generation TKIs, particularly Osimertinib, selectively target T790M-mutant EGFR and have demonstrated improved efficacy and tolerability, becoming a key treatment option. Ongoing research is exploring fourth-generation inhibitors such as JIN-A02 designed to overcome resistance associated with the C797S mutation.
In Colorectal Cancer (CRC), EGFR-targeted therapy is an important treatment strategy for patients with microsatellite-stable and RAS wild-type metastatic disease. Monoclonal antibodies such as Cetuximab and Panitumumab inhibit ligand-induced EGFR activation and downstream signaling pathways, and are commonly combined with chemotherapy regimens such as FOLFOX or FOLFIRI to improve clinical outcomes. Their benefit is restricted to RAS wild-type tumors, and targeted combinations such as Encorafenib plus cetuximab are approved for patients with BRAF V600E–mutant disease following prior therapy. Emerging approaches, including combinations with KRAS G12C inhibitors and biomarker-guided rechallenge strategies, are further expanding treatment options.
In Head and Neck Cancer, treatment typically involves chemotherapy, targeted therapy, and radiation therapy. EGFR inhibition represents a key targeted strategy, with Cetuximab approved for use in combination with radiation therapy or chemotherapy in certain patients. Cetuximab binds to the extracellular domain of EGFR, blocking ligand binding and inducing antibody-dependent cellular cytotoxicity, thereby inhibiting tumor growth and survival. EGFR TKIs such as Erlotinib and Afatinib have also been investigated for head and neck cancers, although monoclonal antibody–based EGFR inhibition remains the primary targeted approach in this setting.
Further details related to country-based variations are provided in the report.
Epidermal Growth Factor Receptor (EGFR) Inhibitor Unmet Needs
The section “unmet needs of Epidermal Growth Factor Receptor (EGFR) Non-small Cell Lung Cancer (NSCLC)” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Limited options after first-line Osimertinib
- CNS Metastases and Limited Brain Penetration
- Limited Access and Affordability
- Therapy Gaps after Targeted Therapy Progression, and others…..
Note: Comprehensive unmet needs insights in EGFR NSCLC and their strategic implications are provided in the full report.
Epidermal Growth Factor Receptor (EGFR) Inhibitor Epidemiology
Key Findings from EGFR targeted cancer Epidemiological Analysis and Forecast
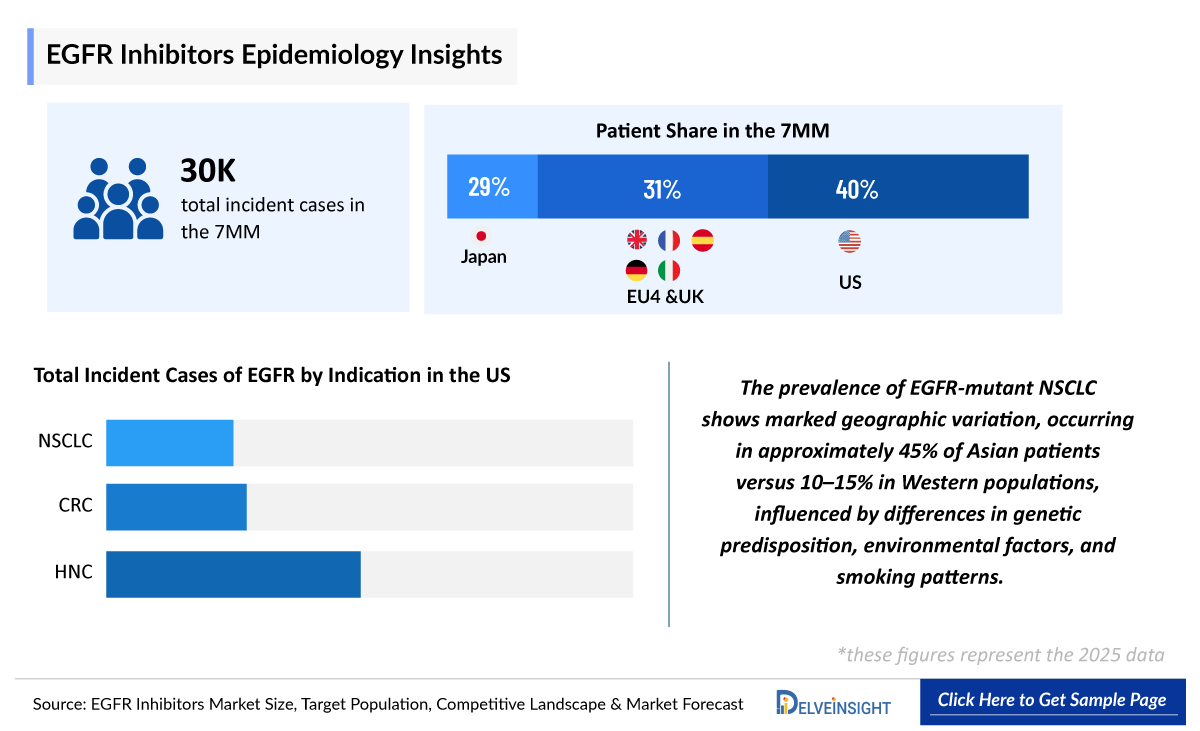
- According to DelveInsight’s estimates, the total number of incident cases of NSCLC in the 7MM was nearly 115,000 cases in 2025 and is projected to increase during the forecasted period.
- The total number of incident cases of NSCLC in the United States was nearly 30,000 in 2025.
- In the 7MM, the US accounted for the highest incident cases of EGFR mutated cancers in 2025, with around 118,790 cases; these numbers are expected to increase during the forecast period.
- In 2025, the EGFR-expressing incident cases of selected indications in Japan included 55,000 cases of NSCLC, 33,000 cases of CRC, and 17,000 cases of HNC.
- In the EU4 and the UK, Germany accounted for the highest incident cases of EGFR mutated cancers with approximately 35,000 cases reported in 2025.
Epidermal Growth Factor Receptor (EGFR) Inhibitors Drug Analysis & Competitive Landscape
The EGFR inhibitors drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I/II–II clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the EGFR inhibitor treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the EGFR inhibitor therapeutics market.
Approved Therapies for EGFR inhibitor
Osimertinib (TAGRISSO): AstraZeneca
Osimertinib is a prescription medicine for adults with NSCLC with abnormal EGFR genes. It is used to prevent recurrence after surgery, as a first-line treatment for metastatic NSCLC, or when previous EGFR TKI treatments have failed. Osimertinib is a kinase inhibitor that targets mutant EGFR forms (T790M, L858R, exon 19 deletions) at lower concentrations than wild-type EGFR. In November 2015, it was initially approved 80mg once-daily tablets for the treatment of patients with metastatic EGFR T790M mutation-positive NSCLC. In February 2024, the FDA approved osimertinib with platinum-based chemotherapy for patients with locally advanced or metastatic NSCLC whose tumors have EGFR exon 19 deletions or exon 21 L858R mutations.
Note: Detailed marketed therapies assessment will be provided in the final report.
EGFR Inhibitor Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region (First approval) |
|
Osimertinib (TAGRISSO) |
AstraZeneca |
EGFR T790M mutation-positive metastatic NSCLC |
Small molecule |
EGFR-TKi |
Oral |
US: Conditional approval: November 2015, Full approval: March 2017 |
|
Sunvozertinib (ZEGFROVY) |
Dizal Pharmaceutical |
EGFR exon 20 insertion NSCLC |
Small molecule |
Irreversible EGFR inhibitor |
Oral |
US: July 2025 |
|
Erbitux |
Eli Lilly and Company/ Bristol Meyer Squibb/Merck KGaA |
EGFR-expressing, mCRC |
Monoclonal Antibody |
EGFR antagonist |
Intravenous |
US: 2004 EU: 2004 JP: 2008 |
Epidermal Growth Factor Receptor (EGFR) Inhibitors Pipeline Analysis
Zipalertinib: Cullinan Oncology/Taiho Pharmaceutical
Zipalertinib (CLN-081/TAS6417) is a novel, orally bioavailable, irreversible EGFR inhibitor that, based on preclinical models, selectively and potently targets cells expressing EGFRex20ins mutations while relatively sparing cells expressing wild-type EGFR to avoid the toxicities associated with inhibition of wild-type EGFR. This was rationally designed with a distinct chemical scaffold to be highly selective for mutant vs. wild-type EGFR and to avoid inhibiting the closely related receptor human epidermal growth factor receptor 2 (HER2). Zipalertinib demonstrates the potential to become a new standard of care to treat non-small cell lung cancer harboring EGFRex20ins mutations.
EGFR Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Zipalertinib (CLN-081) |
Cullinan Oncology/ Taiho Pharmaceutical |
III |
EGFR Exon 20 ins NSCLC |
Oral |
Irreversible EGFR inhibitor |
2027 |
|
Firmonertinib |
ArriVent BioPharma |
III |
EGFR Exon 20 ins NSCLC, EGFR PACC NSCLC |
Oral |
EGFR inhibitor |
Information is available in the full report |
|
Ivonescimab (SMT112) |
Akeso Biopharma/Summit Therapeutics |
III |
Advanced NSCLC/EGFR mutant advanced NSCLC |
IV infusion |
PD-1/VEGF inhibitor |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Epidermal Growth Factor Receptor (EGFR) Inhibitors Key Players, Market Leaders and Emerging Companies
- Cullinan Oncology
- Taiho Pharmaceuticals
- ArriVent BioPharma
- Akeso Biopharma
- Summit Therapeutics
- Pfizer
- Daiichi Sankyo
- AstraZeneca
- Merck
- Kelun-Biotech
- CSPC Pharmaceutical
- Shanghai JMT-Bio
- Bristol-Myers Squibb, and others
Recent Developments in Epidermal Growth Factor Receptor (EGFR) Inhibitors Clinical Trials
- In February 2026, Bicara Therapeutics Inc. presented preliminary safety and efficacy data from an exploratory Phase Ib expansion cohort evaluating 2000mg of ficerafusp alfa every other week (Q2W) in combination with pembrolizumab in first-line (1L) human papillomavirus (HPV)-negative recurrent/metastatic (R/M) head and neck squamous cell carcinoma (HNSCC). The expansion cohort data, which explore a higher dose and less-frequent dosing regimen of ficerafusp alfa, were highlighted in a plenary presentation at the 2026 Multidisciplinary Head and Neck Cancers Symposium (MHNCS).
- In January 2026, the US FDA accepted for filing Summit’s Biologics License Application (BLA) for the HARMONi Phase III trial and assigned a Prescription Drug User Fee Act (PDUFA) goal action date of November 14, 2026.
- In November 2025, a New Drug Application (NDA) was submitted for zipalertinib for the treatment of EGFR exon 20–mutant NSCLC, with completion and priority review planned for Q1 2026, based on data from the REZILIENT1 Phase I/II study.
- Silvertinib is being evaluated for 1L NSCLC, with Phase II DOR/PFS data expected in Q2 2026 and subsequent FDA feedback. Final Phase II data in 2L/3L NSCLC are also expected in Q2 2026, and the drug has received FDA Fast Track Designation (FTD) for C797S+ patients while partnering options for pivotal development are being explored.
Epidermal Growth Factor Receptor (EGFR) Inhibitor Market Outlook
The market outlook for EGFR inhibitors remains strong, driven by the growing adoption of precision oncology and the expanding role of targeted therapies across multiple tumor types. In EGFR-mutated NSCLC, third-generation inhibitors such as Osimertinib have established a dominant position in the first-line setting due to improved survival outcomes and strong central nervous system activity. However, the emergence of resistance mechanisms is accelerating the development of next-generation inhibitors, bispecific antibodies, and combination regimens aimed at addressing post-progression treatment gaps. In parallel, EGFR-targeted monoclonal antibodies including Cetuximab and Panitumumab continue to maintain clinical relevance in colorectal and head and neck cancers. With increasing focus on uncommon EGFR mutations, novel therapeutic modalities, and expanding patient identification through molecular testing, the EGFR inhibitor market is expected to witness sustained growth and continued innovation in the coming years.
Treatment of EGFR-mutant NSCLC has improved with targeted EGFR TKIs over the past two decades. First-generation TKIs (erlotinib, gefitinib), second-generation TKIs (afatinib, dacomitinib), and the third-generation TKI (osimertinib) have progressively improved response and survival compared with chemotherapy. Osimertinib is also effective against the T790M resistance mutation; however, drug resistance still develops and optimal treatment after TKI failure remains challenging.
Osimertinib remains the global standard of care for EGFR-mutant NSCLC, supported by strong overall survival (OS) and progression-free survival (PFS) data and broad approvals across disease stages. Amivantamab is emerging as a key competitor, expanding beyond EGFR exon 20 mutations through combination regimens with lazertinib and chemotherapy. The landscape is becoming more competitive with new entrants such as Datopotamab deruxtecan, Sunvozertinib, and Aumolertinib. Real-world data from the 7MM show that EGFR TKIs dominate treatment, with osimertinib widely used as first-line therapy, while chemotherapy and immunotherapy remain options in later lines or for rare mutations (e.g., exon 20 insertions).
Cetuximab (Erbitux), an anti-EGFR IgG1 monoclonal antibody, is expected to maintain a stable yet mature market position across metastatic colorectal cancer (mCRC) and head and neck cancers, supported by its long-standing clinical use, strong evidence base, and approvals from the U.S. Food and Drug Administration, European Medicines Agency, and Pharmaceuticals and Medical Devices Agency. In mCRC, its role remains prominent in biomarker-selected populations, particularly in EGFR-expressing and RAS wild-type tumors, with additional momentum from combination regimens involving targeted agents such as Encorafenib for BRAF V600E-mutated disease and Adagrasib for KRAS G12C-mutated tumors. Similarly, in head and neck cancers, cetuximab continues to see steady utilization in combination treatment regimens and in patients unsuitable for certain systemic or immunotherapy options. Despite sustained clinical demand across major markets, overall market growth is expected to remain moderate due to its mature lifecycle, biosimilar competition, and the increasing availability of newer targeted and immunotherapy treatments.
EGFR exon 20 insertion NSCLC is difficult to treat with standard TKIs. Amivantamab was the main approved therapy after mobocertinib was withdrawn. In 2025, Sunvozertinib was approved, increasing competition, while TKIs like Furmonertinib and Zipalertinib are in development. Emerging ADCs and next-generation therapies are further intensifying competition in the EGFR NSCLC market.
Overall, the launch of first-in-class therapies, improved genetic testing, and rising disease awareness are expected to drive steady growth in the 7MM EGFR inhibitor market from 2022–2036, with strong commercial implications for both marketed products and emerging pipelines.
- In 2025, the United States held the largest market share for EGFR inhibitors among the 7MM, accounting for approximately 60% of the total market.
- Germany shows consistent growth in NSCLC revenues, reflecting steady adoption of EGFR-targeted therapies and increasing treatment penetration. HNC revenues accelerate significantly after the late 2020s, suggesting entry of new targeted or biologic therapies that drive market expansion. Meanwhile, CRC revenues grow modestly, highlighting a stable but limited-growth market due to the presence of established treatment options.
- Cetuximab continues to dominate the EGFR inhibitor market in Head and Neck Cancer, primarily because it remains the only widely approved EGFR-targeted therapy for this indication. As a result, it is extensively utilized in clinical practice both as a monotherapy and in combination with chemotherapy or platinum-based regimens, making it the cornerstone of EGFR-directed treatment strategies for patients with head and neck cancer.
- In 2036, among all the therapies for EGFR NSCLC, the highest revenue is estimated to be generated by amivantamab (RYBREVANT) ± lazertinib (LAZCLUZE) followed by osimertinib (TAGRISSO), in the US.
- The entry of late-stage candidates such as zipalertinib, furmonertinib is expected to intensify competition in the EGFR NSCLC treatment landscape during the latter forecast period.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in EGFR Inhibitor (2022–2036 Forecast)
- The EGFR Inhibitor market comprises monoclonal/bispecific antibody, ADC, and small molecules, each targeting different aspects of EGFR inhibitor.
- Small molecules: Small-molecule therapies in development for EGFR-mutant NSCLC include zipalertinib (CLN-081) by Cullinan Oncology/Taiho Pharma, firmonertinib by ArriVent BioPharma, and LP-300 by Lantern Pharma.
- ADCs: Key ADCs in the pipeline include PF-08046054 (Pfizer), patritumab deruxtecan (Daiichi Sankyo/AstraZeneca), sacituzumab tirumotecan (MK-2870) (Merck/Kelun-Biotech), SYS6010 (CSPC Pharmaceutical), izalontamab brengitecan (BMS-986507) (Bristol-Myers Squibb), and telisotuzumab adizutecan (ABB-400) (AbbVie).
- Small molecules and ADCs defines the core innovation landscape, with monoclonal and bispecific antibodies currently commercially validated and small molecules driving pipeline growth.
Epidermal Growth Factor Receptor (EGFR) Inhibitor Drug Uptake
This section focuses on the uptake rate of potential EGFR drugs expected to be launched in the Epidermal Growth Factor Receptor (EGFR) Inhibitor market during the forecast period (2026–2036). The analysis covers the EGFR inhibitor drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
The uptake of therapies in EGFR NSCLC is expected to vary based on clinical positioning, mechanism of action, and stage of development. Adoption of amivantamab (RYBREVANT) + chemotherapy and amivantamab (RYBREVANT) + lazertinib (LAZCLUZE) in EGFR NSCLC is expected to occur at a moderate uptake rate, supported by their targeted approach, favorable clinical data, and increasing integration into earlier lines of therapy.
The adoption of emerging therapies in EGFR-mutant NSCLC is expected to vary based on clinical maturity, combination strategies, and differentiation from existing standards of care. Furmonertinib/firmonertinib may achieve relatively faster uptake due to encouraging clinical data and potential positioning in targeted therapy settings. In contrast, pamvatamig (MCLA-129) ± osimertinib and telisotuzumab adizutecan (Temab-A) + TAGRISSO are likely to experience gradual uptake, reflecting their earlier clinical positioning and the need for stronger comparative evidence against established EGFR TKIs.
Detailed insights of emerging therapies' drug uptake is included in the report
Market Access and Reimbursement of Approved therapies in Epidermal Growth Factor Receptor (EGFR) targeted cancers
The Epidermal Growth Factor Receptor (EGFR) Inhibitor market report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
Epidermal Growth Factor Receptor (EGFR) Inhibitor Therapies Price Scenario & Trends
Pricing and analogue assessment of EGFR inhibitor therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most approproiate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of EGFR NSCLC Approved Drugs
In the first-line setting, treatment costs vary significantly, with platinum-based chemotherapy estimated at USD 8,747, first- and second-generation TKIs at USD 27,943, and osimertinib reaching approximately USD 212,580, reflecting the higher cost associated with targeted therapies.
Industry Experts and Physician Views for Epidermal Growth Factor Receptor (EGFR) Inhibitors
To keep up with EGFR Inhibitor market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the EGFR inhibitors emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in EGFR NSCLC, including MD, Ph.D, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 15+ KOLs to gather insights at country level. Centers such as the Ohio State University, Norris Comprehensive Cancer Center, and University of Southern California, etc. were contacted. Their opinion helps understand and validate current and emerging EGFR NSCLC therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in EGFR NSCLC.
What are the Latest KOL Views on Epidermal Growth Factor Receptor (EGFR) Inhibitor Market? | |
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Patients with EGFR Exon 20 insertion mutations need more effective therapies. Although second-line agents are approved for this population, they are associated with toxicities, and the mechanism of disease progression on these agents is unclear.” |
|
Germany |
“Even in a contemporary era with prevalent osimertinib use, the baseline and longitudinal risk of BM development was high. The ongoing risk of developing BM, together with the associated survival detriment, argues for routine surveillance of the brain through magnetic resonance imaging for patients with EGFR-mutated NSCLC, which is not currently included in the guidelines.” |
Epidermal Growth Factor Receptor (EGFR) Inhibitor Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of EGFR Inhibitors, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Epidermal Growth Factor Receptor (EGFR) Inhibitor Market Report
- The Epidermal Growth Factor Receptor (EGFR) Inhibitor market report covers a segment of key events, an executive summary, a descriptive overview of Epidermal Growth Factor Receptor (EGFR) Non-small Cell Lung Cancer (NSCLC), CRC and HNC, and EGFR inhibitors explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Epidermal Growth Factor Receptor (EGFR) inhibitors market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Epidermal Growth Factor Receptor (EGFR) Inhibitor market report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Epidermal Growth Factor Receptor (EGFR) inhibitors market.
Epidermal Growth Factor Receptor (EGFR) Inhibitor Market Report Insights
- EGFR Inhibitor Patient Population Forecast
- EGFR Inhibitor Therapeutics Market Size
- EGFR Inhibitor Pipeline Analysis
- EGFR Inhibitor Market Size and Trends
- EGFR Inhibitor Market Opportunity (Current and forecasted)
Epidermal Growth Factor Receptor (EGFR) Inhibitor Market Report Key Strengths
- Epidemiology-based (Epi-based) Bottom-up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year forecast
- EGFR Inhibitor Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- EGFR Inhibitor Treatment Addressable Market (TAM)
- EGFR Inhibitor Competitive Landscape
- EGFR Inhibitor Major Companies Insights
- EGFR Inhibitor Price Trends and Analogue Assessment
- EGFR Inhibitor Therapies Drug Adoption/Uptake
- EGFR Inhibitor Therapies Peak Patient Share Analysis
Epidermal Growth Factor Receptor (EGFR) Inhibitor Market Report Assessment
- EGFR Inhibitor Current Treatment Practices
- EGFR Inhibitor Unmet Needs
- EGFR Inhibitor Clinical Development Analysis
- EGFR Inhibitor Emerging Drugs Product Profiles
- EGFR Inhibitor Market Attractiveness
- EGFR Inhibitor Qualitative Analysis (SWOT and conjoint analysis)
Frequently Asked Questions from Epidermal Growth Factor Receptor (EGFR) Inhibitor Market Report
EGFR Inhibitor Market Insights
- What was the EGFR Inhibitor market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of EGFR Inhibitor?
- What impact will patent expiry have on the EGFR therapy market?
- What are the disease risks, burdens, and unmet needs of EGFR Inhibitor? What will be the growth opportunities across the 7MM concerning the patient population with EGFR Inhibitor?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of EGFR Inhibitor? What are the current guidelines for treating EGFR Inhibitor in the US, Europe, and Japan?
Reasons to Buy the EGFR Inhibitor Market Report
- The EGFR Inhibitor market report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the EGFR Inhibitor market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing EGFR Inhibitor market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming EGFR Inhibitor companies in the EGFR Inhibitor market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming EGFR Inhibitor companies can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.