Overcoming the prevailing unmet needs in Beta-Thalassemia Market
May 08, 2020
The current Beta-thalassemia market is experiencing the emergence of gene therapies as a one-time cure for the patients.
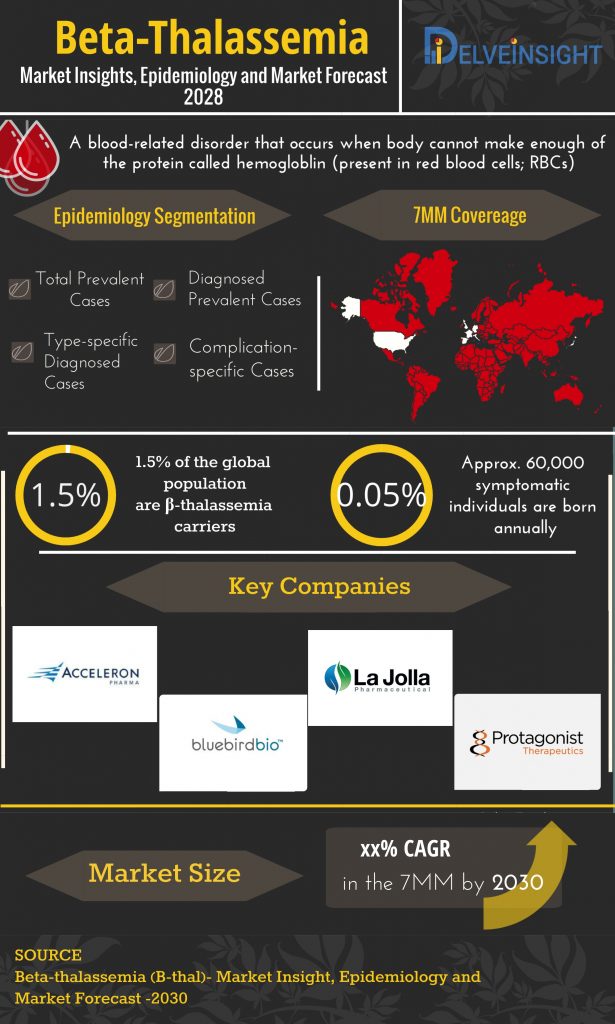
Beta thalassemia is relatively a common heterogeneous autosomal recessive hereditary blood disorder. It is a common form of hemoglobinopathy characterized by ineffective erythropoiesis leading to reduced or absent β-globin chain synthesis, severe anemia and extensive erythroid expansion.
Each year thousands of infants are born afflicted with beta-thalassemia. Beta thalassemia is majorly prevalent in people from Mediterranean countries, North Africa, the Middle East, India, Central Asia, South Africa and Southeast Asia.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Taysha Acquires Rare Disease Gene Therapy For $5.5 M; AZ’s Farxiga Fails In COVID-19 Study; Sanof...
- Mylan closing Illinois plant; Insys spends $24M; Roche’s Ocrevus; Greece’s corruption prose...
- Watershed Moment for Cell Therapies and Complicated Journey of Gene Therapies in Japan
- Bayer eyes AskBio’s Gene Therapies; Novartis’ Iptacopan; Forma’s IDH1-Mutated AML The...
- Pfizer succeeds COVID-19 vaccine safety milestone; Sanofi snags speedy review for BTK drug; Autoi...
According to the study by Galanello, R., & Origa, R. (2010), The total annual incidence of symptomatic individuals was estimated at 1 in 100,000 throughout the world and 1 in 10,000 people in the European Union. Moreover, according to the Thalassemia International Federation, about 200,000 patients with thalassemia major are alive and registered as receiving regular treatment around the world.
Beta thalassemia, based on genetic characteristics and clinical features expressed by the patients, is categorized into three major sub-types- minor (carrier), intermedia, and major. The pharmacological treatment option available in the beta-thalassemia marker is regular blood transfusion, absence or failure of which is associated with a high mortality rate. Blood transfusion therapy and oral iron chelation therapy have impacted the beta-thalassemia market positively, offering relief from a once fatal disease. Generally, blood-transfusion begin early in childhood. The aim of transfusion is mainly to suppress erythroid expansion. The therapy aims to diminish the symptoms of anemia.
However, the available therapies are symptomatic and last life-long. Blood transfusion therapy at regular intervals of confines patients and also increases the risk of iron overload. In addition to iron overload, transfusion therapy also involves the risk of developing red blood cell antibodies thus finding a donor that suits the best becomes a tedious yet imperative task. It is also to be noted that therapy altogether depends on the availability of a donor. At some places, the demand is met; however, in some geographies, especially, middle- and low-income countries, the demand ousts the supply. Often times, the donors do not fall under the voluntarily non-remunerated category that further increases the financial burden on the patients and their care-takers.
Another major consequence of undergoing repetitive blood transfusion is an excessive build-up of iron since the body lacks an effective excretory pathway for iron removal. However, for this, Iron chelation therapy is used. There are some iron chelators used in iron chelation therapies namely; Deferoxamine (DFO), Deferiprone (DFP) and Deferasirox (DFX). Desferal (deferoxamine mesylate USP), Exjade (Deferasirox) are the candidates of Novartis, whereas Ferriprox (deferiprone) is manufactured by Apotex.
At present, the only cure that appears potential enough in beta-thalassemia market and can be considered a definitive cure is bone marrow transplantation. Besides these, cord blood transplant is considered for parents who already have a child with beta-thalassemia and later identify an HLA-compatible fetus in a subsequent pregnancy.
The current Beta-thalassemia market is experiencing the emergence of gene therapies as a one-time cure for the patients. Key players, such as Bluebird Bio, Protagonist Therapeutics, Acceleron Pharma, Sangamo Therapeutics, Sanofi, Fulcrum Therapeutics, Orchard Therapeutics and others are proactively working towards strengthening beta-thalassemia market. The launch of various multiple-stage pipeline products will significantly revolutionize the β-thal market dynamics with the continuous efforts in research and development.
LentiGlobin BB305 (Bluebird Bio) – a lentiviral vector, Luspatercept or ACE-536 (Acceleron) – a first-in-class erythroid maturation agent, and Sotatercept or ACE-011 (Acceleron) – a biologic drug, PTG-300 (Protagonist Therapeutics) – an injectable hepcidin mimetic peptide are some of the emerging therapies in the pipeline for the beta-thalassemia market. Last year Fulcrum announced progress in its plan to develop FTX-6058 for the potential treatment of sickle cell disease and beta-thalassemia. Similarly, Sangamo has recently presented promising results of its cell therapy ST-400, developed in partnership with Sanofi for beta-thalassemia. Along these lines, Orchard Therapeutics presented positive results from the proof-of-concept study of its gene therapy – OTL-300 in transfusion-dependent patients with beta-thalassemia.
Besides a robust and novel-drug rich pipeline, improved diagnosis methodologies, better awareness, increased R&D, and incremental healthcare spending shall veer the competitive landscape for the Beta-thalassemia (β-thal) market during the forecast period, 2019–2028.
Downloads
Article in PDF
Recent Articles
- Why should every company working on CAR-T therapy attend CAR TCR Summit Europe?
- Notizia
- COVID19 pipeline advances as Novartis begins phase III trial of hydroxychloroquine; and advanceme...
- Can Dupixent Be A Gamechanger In The Atopic Dermatitis Treatment Landscape?
- Novartis eyes; Pfizer considers cashing; Mylan pushes to seal $465M; Valeant with Takeda for deal