Can Cannabinoids be an Effective Medicinal Substance?
Mar 16, 2022
Table of Contents
Cannabidiol (CBD) is one of the 120 cannabinoids present naturally in cannabis plants. After tetrahydrocannabinol (THC), Cannabidiol is the second most prevalently available cannabinoid. Cannabidiol, an important component of medicinal marijuana, is either directly from the hemp plant or synthesized in a laboratory. Cannabidiol in a prescription form is used to treat seizure disorders such as Epilepsy and others. Cannabidiol is also used to treat Anxiety, Pain, Dystonia, Parkinson’s disease, Crohn’s disease, and various other diseases, although no clear scientific evidence supports its uses.
The 2018 Farm Bill removed all hemp-derived products, including Cannabidiol, from the Controlled Substances Act, which criminalizes drug possession in the United States. In essence, this implies that Cannabidiol is legal if derived from hemp but illegal if derived from cannabis (marijuana) — even though they are the same molecule.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Gut bacteria can stop cancer drugs from working
- Potential of Digital therapeutics and increasing CVDs
- Human cells reprogrammed to create insulin in diabetic mice
- Machine Learning Models to Help Predicting Cancer Symptoms, Plan Treatment
- REVIAN RED System Delivers Positive Results, Insulet’s Omnipod Provides Notable Improvement in Ty...
Types of Cannabidiol Products
There are different types of Cannabidiol products available based on its method, even though the effect tends to be the same. The Cannabidiol types include:
- Cannabidiol oils and tinctures– They are often made by infusing Cannabidiol with liquids such as glycerin, oil, or alcohol as a basis. It can be consumed sublingually, due to which there is a rapid onset of the effect.
- Cannabidiol edibles– comprise the extracts mentioned above but in a culinary form. Cannabidiol edibles are available in the form of beverages and foods such as teas and chocolate. Cannabidiol gummies are a popular type of CBD edible. They are taken orally and may take longer to show an effect.
- Capsules- Cannabidiol-containing pills and soft gels are available that can be taken orally but have a longer onset period than Cannabidiol oils (CBD oils) and creams.
- Cannabidiol Vaporizers– Vaporizers can deliver Cannabidiol extracts by inhalation for immediate impact. However, vaping has additional risks, including lung damage. Sublingual extracts applied under the tongue may give some of the same fast effects as vaporizers without the danger of vaping-related lung harm.
- Creams and lotions- Cannabidiol is also available in the form of topical oils, creams, and lotions. Some people may feel the effects immediately, whereas others may not notice any effects for several hours.
The classification of Cannabidiol products based on the amount of Cannabidiol extract present is as follow:
- Cannabidiol isolates- Cannabidiol isolate oil exclusively contain Cannabidiol from the cannabis plant. There are no additional chemicals present, such as THC. CBD-only products can assist a person in determining the efficacy of Cannabidiol without influence from other substances and components.
- Broad-spectrum Cannabidiol- Broad-spectrum Cannabidiol products contain other chemicals and cannabinoids found in the plant, such as CBC, CBN, or terpenes like myrcene, limonene, or pinene
- Full-spectrum Cannabidiol– Those that are full-spectrum Cannabidiol products include the same chemicals as products that are broad-spectrum. They, however, frequently include trace amounts of THC, usually less than 0.3% as required by federal law.
Cannabinoids Benefits and Uses
Cannabinoids contain anti-inflammatory, anticonvulsive, analgesic, anxiolytic, sedative, and antipsychotic characteristics due to their interaction with the body’s endocannabinoid system. According to the research of CBD users published in Cannabis and Cannabinoid Research, around 62% of participants used Cannabinoids to treat medical problems. Depression, Anxiety, and Pain were the most frequent among these disorders. The significant scientific evidence establishes its effectiveness in treating some epileptic disorders, including Dravet syndrome and Lennox-Gastaut Syndrome (LGS), that do not respond to antiseizure drugs.
Cannabinoids may also potentially be helpful in the treatment of Insomnia, Addiction, Chronic Pain, and Multiple Sclerosis (MS). The scientific study supporting these alleged medicinal advantages of Cannabinoids is still in its initial stages. Anecdotal data suggest that persons who have taken Cannabinoids have experienced relief from a wide range of ailments. Cannabinoids can be a significant medicinal compound due to their many beneficial properties.
The FDA recognizes the potential advantages of Cannabinoids, but the agency is quick to point out that further study is needed. Epidiolex is the first FDA-approved Cannabinoids drug used to treat severe Epilepsy.
Cannabinoids for Various Indications
Osteoarthritis- Osteoarthritis is a form of non-inflammatory arthritis caused by the deterioration of cartilage, the protective bone cushioning found at joints. It affects millions of individuals worldwide. Although Osteoarthritis may affect any joint, it most usually affects the hands, knees, hips, and spine. Cannabidiol oil and topical Cannabidiol products, due to their anti-inflammatory and analgesic qualities, can aid in symptom alleviation in common kinds of arthritis, such as Osteoarthritis and Rheumatoid Arthritis. A synthetic transdermal CBD gel trial in adults with Knee Osteoarthritis reported that some participants had considerable pain relief, with males benefiting more than women. CBD has also been demonstrated to have the ability to reduce pain and nerve damage in Osteoarthritis patients. Cannabinoids may potentially provide indirect advantages to Osteoarthritis patients, such as improving sleep and mood-related issues.
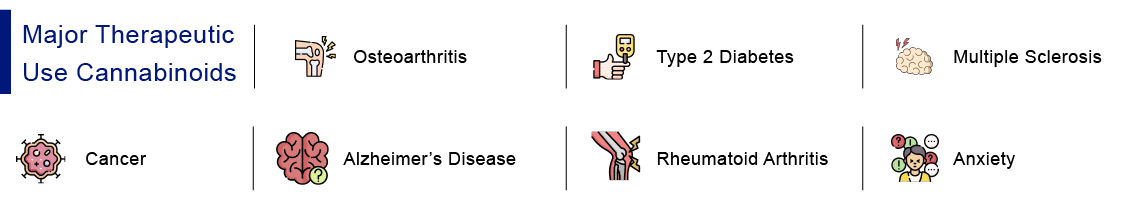
Type 2 Diabetes- Most trials on CBD’s impact on diabetes have been conducted on mice or rats. This is a concern since there may be a difference in effectiveness between animals and humans. In one study, researchers tried Cannabidiol on mice with decreased blood flow to the brain, which is a complication of diabetes in some humans. They discovered that Cannabidiol for diabetes can:
- Reduce hyperglycemia
- Lower amounts of cholesterol
- Increased insulin production
Cannabidiol has also been shown in animal experiments to reduce swelling and discomfort caused by nerve degeneration, which affects the hands and feet of diabetic patients. Cannabidiol may also be able to lower insulin resistance and boost gut hormone levels. However, it has shown no improvement in blood sugar and lipid levels in persons with Type 2 Diabetes. Another concern associated with CBD is the lack of FDA regulation, even though it’s available in many forms, from liquid drops to capsules to vapes.
Cancer– There are currently no FDA-approved Cannabidiol medications used for treating cancer. But there are ongoing studies that state that Cannabidiol could relieve some of the cancer side effects and its treatment, such as pain, insomnia, anxiety, or nausea. Some studies have shown that Cannabidiol inhibits the growth of cancer cells in mice with Lung Cancer or Colon Cancer. Together with THC, it can kill glioblastoma cancer cells. However, no studies have been conducted on people with cancer. According to ASCO guidelines, cannabinoids may be prescribed for chronic pain management, provided it is legal in the state. The guidelines, however, acknowledge a lack of evidence to support the use of cannabinoids to prevent nausea and vomiting in cancer patients. There are presently 2 synthetic cannabis medications approved by FDA namely, nabilone (Cesamet) and dronabinol (Marinol or Syndros), to treat nausea and vomiting related to chemotherapy. It is also important to note that some studies have shown that CBD might interfere with how the body processes cancer drugs, called a drug interaction, and this might make cancer treatments more toxic or less effective. Hence, the safety and efficacy of CBD for people with cancer still has to be proven in large.
Alzheimer– Alzheimer’s is a type of dementia that worsens with time. Alzheimer’s disease is characterized by memory, language, and cognitive loss. According to 2019 research, Cannabidiol may effectively treat and prevent Alzheimer’s disease since Cannabidiol components may reduce specific symptoms such as behavioural abnormalities and memory loss. Despite the possibility of alleviating certain symptoms, Cannabidiol may not be able to alleviate the effects of Alzheimer’s as the disease advances. Furthermore, it showed that combining CBD and THC might be more beneficial than using either one alone. However, research is currently limited, and additional human trials are required to determine whether Cannabidiol can treat Alzheimer’s.
Rheumatoid Arthritis– Rheumatoid arthritis is a form of non-inflammatory arthritis. It is an autoimmune disorder in which the immune system destroys its own joint tissue by mistake. The ECS regulates important functions such as pain, inflammation and immune function, and it is responsible for maintaining a delicate state of equilibrium known as homeostasis. By interacting with the body’s endocannabinoid system (ECS), Cannabidiol can alleviate arthritic pain. In a trial of Rheumatoid Arthritis patients, Sativex (a drug with a 1:1 ratio of THC to CBD) relieved joint discomfort and sleep. A case study found that utilizing Cannabidiol oil helped a 50-year-old lady with Rheumatoid Arthritis and two other autoimmune conditions improve her pain, mobility, mood, and inflammation.
Epilepsy– Epilepsy is a central nervous system (neurological) disorder characterized by abnormal brain activity, resulting in seizures or episodes of odd behaviour, sensations, and, in some cases, loss of awareness. Cannabidiol’s medicinal use is most developed in the treatment of Epilepsy. Cannabidiol has been shown in clinical trials to be a successful therapy for at least certain types of Epilepsy. Epidiolex is the first Cannabinoids medicine to be licensed by the FDA for the treatment of severe Epilepsy. CBD’s usage for this purpose is based on a number of animal studies dating back to the 1970s. Cannabidiol has been studied in epilepsy patients based on this research. The FDA has authorized GW Pharmaceuticals’ Epidiolex to treat seizures caused by Dravet syndrome, Lennox-Gastaut syndrome, or tuberous sclerosis.
Anxiety– An increasing amount of evidence shows that Cannabidiol may help reduce anxiety, the most prevalent mental health issue. Cannabidiol interacts with the cannabinoid type 1 receptor (CB1R), the serotonin 5-HT1A receptor, and other brain receptors that control fear and anxiety-related behaviours. Cannabidiol has been shown in preclinical and clinical studies to be a helpful therapy for a variety of anxiety disorders, including:
- Generalized Anxiety Disorder (GAD)
- Post-traumatic stress disorder (PTSD)
- Panic disorder (PD)
- Obsessive-compulsive disorder (OCD)
- Social anxiety disorder (SAD)
Cannabidiol is seen to be a successful monotherapy or adjunct therapy for the treatment of anxiety disorders. However, the trials did demonstrate a significant variance in Cannabidiol dosages, ranging from 6-400 mg per dose. This diversity emphasizes the need for further high-quality, large-scale clinical trials to aid in developing standardized dose and clinical use recommendations for Cannabidiol.
Ongoing Clinical Trials
- Epidiolex, manufactured by GW Pharmaceuticals, has shown promising results in Phase III studies in the United Kingdom for Dravet and Lennox-Gastaut syndromes, both of which are treatment-resistant seizure disorders.
- Arvisol, an oral tablet containing pure Cannabidiol created by Echo Pharmaceuticals, is being used to treat various neurological disorders, including schizophrenia and Epilepsy. Arvisol is currently in the early stages of clinical studies.
- Zynerba Pharmaceuticals is working on a Cannabidiol gel called Zygel. The drug is intended to treat Fragile X syndrome, adult seizures, and encephalopathies. The formulation is currently in open-label Phase II testing for Fragile X syndrome.
- STI Pharmaceuticals has developed a crystalline powder of pure synthetic CBD with multiple doses that have been evaluated in a Phase II study for its effects on marijuana-induced subjective effects in an oral capsule formulation.
- INSYS Pharmaceuticals (United States) has created an oral solution containing pure CBD. It is now in Phase 2 trials for childhood absence seizures (20-40 mg) and a Phase 3 trial for infantile spasm-type seizures in combination with vigabatrin.
- PhytoTech Therapeutics (Tel Aviv, Israel) is working on an oral formulation (PTL101) with pure CBD contained in gelatin matrix pellets. This product has undergone phase 1 testing (10 to 100 mg) and was found to have considerably higher bioavailability when compared to a CBD-containing reference product.
Recent Developments in the Cannabidiol Market
- In February 2022, Cann Group has starts recruitment for the Phase III clinical trial for its CBD candidate (Satipharm CBD) for sleep issues/sleep disturbances. Satipharm CBD formulation will be evaluated in nearly 212 patients against placebo for over four weeks. The trial results are expected to be released later this year.
- On February 2022, MJH Life Sciences announced the acquisition of Cannabis Science Conference.
- Recently, in March 2022, the European Commission has granted orphan drug designation to Zynerba Pharmaceuticals’ Zygel. Zygel, is a transdermal cannabidiol gel candidate developed to treat behavioural symptoms in people with fragile X syndrome. Zygel is currently in Phase 2/3 trial.
- In one of the latest acquisition update in the Cannabidiol Market, the Toronto-based cannabis company TerrAscend Corp. has anoucned that it is in a move to acquire Gage Growth Corp, which one of the largest Cannabis sellers in Michigan, USA.
- On February 2022, InterCure acquired Israeli Cannabis Company Cann Pharmaceutical, also known as Better for USD 24.7 million.
- On February 23, 2022, Averix Bio, a cannabinoid research & extraction company based in the US, has announces its submission of a Drug Master File (DMF) to the FDA for cannabidiol (CBD) isolate. Averix ingredients are not synthetic but instead made from phytocannabinoid industrial hemp.
Key Companies in the Cannabidiol Market
Globally, some of the key pharma companies actively working in the Cannabidiol Market include Gefion Canada Inc, KGK Science Inc., Defined Research, Radical Science, Formula30A LLC, New Age Ventures LLC, KGK Science Inc., Radical Science Laboratories, Demetrix, Ginkgo Bioworks, Inc., Hyasynth Bio, Creo, Amyris, Zymergen, Canopy Growth Corporation, Willow Biosciences Inc, InMed Pharmaceuticals, CB Therapeutics, Lygos, Purisys, Renew Biopharma, Inc., CBD Oil., Maricann Inc., and several others.
Additionally, pharma giants such as Jazz Pharmaceuticals is currently targeting more than different indications, including Lennox Gastaut Syndrome, Dravet Syndrome, Tuberous Sclerosis Complex, Autism Spectrum Disorder, and Seizure in Participants With Tuberous Sclerosis Complex.

The active participation of the pharma companies towards Cannabidiol as medicine provides high hope for patients suffering from different conditions. Moreover, several of the therapies are in the advanced stage of clinical development that is expected to enter the market in the coming years.
Cannabidiol Side Effects
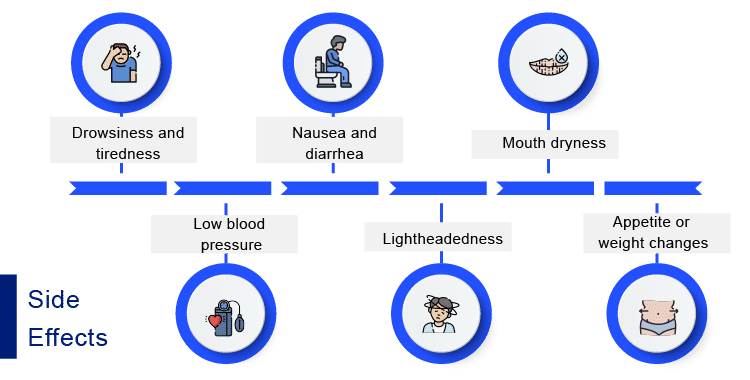
Although CBD can have certain side effects, it is generally regarded as a well-tolerated substance. The adverse cannabidiol effects include Drowsiness and tiredness, nausea and diarrhoea Mouth dryness, Low blood pressure, lightheadedness, appetite or weight changes, among several others.
On the other hand, the bulk of these negative effects are minor and only appear when taking pure CBD at high doses of 300 mg or more. Because most individuals take far smaller doses of CBD and frequently choose whole-plant products that contain many other beneficial chemicals, the chances of having adverse effects from CBD are low. As CBD is difficult to permeate deeply enough into the skin to reach the bloodstream, topical CBD products are much less likely to induce negative effects.
Future of Cannabidiol Market
Cannabinoids is one of the more than 120 cannabinoids found in the cannabis plant. CBD is the most intriguing cannabinoid in terms of pharmacology. Patients with inflammatory, neurodegenerative, and autoimmune diseases, as well as those with epilepsy and cancer, have reported positive therapeutic effects. Current regulations allow the use of CBD-based products in the form of high-concentration cannabidiol oil capsules, powder, and extract.
Based on previous research, an increasing number of countries have changed or are changing their cannabis regulation rules to include CBD as a medicinal product as well as a food supplement. GW Pharmaceuticals’ Epidiolex, the first CBD-based drug, is currently approved in both the United States and Europe for the treatment of some severe forms of Epilepsy. Furthermore, several other companies such as Gefion Canada Inc, KGK Science Inc., and others are also developing CBD for various indications. Moreover, several of the CBD therapies are in the late stages which may soon join the Cannabidiol market.

Frequently Asked Questions (FAQs)
Cannabidiol is the second most prevalently available cannabinoid. Cannabidiol, an important component of medicinal marijuana, is either directly from the hemp plant or synthesized in a laboratory.
Cannabidiol is used to treat different indications such as Osteoarthritis, Type 2 Diabetes, Cancer, Alzheimer’s disease, Rheumatoid Arthritis, Multiple Sclerosis, Anxiety, Epilepsy and others.
Globally, some of the key pharma companies actively working in the Cannabidiol Market include Gefion Canada Inc, KGK Science Inc., Defined Research, Radical Science, Formula30A LLC, New Age Ventures LLC, and several others.
The adverse cannabidiol effects include drowsiness and tiredness, nausea and diarrhoea, Mouth dryness, low blood pressure, lightheadedness, appetite or weight changes, and others.
Cannabinoids may also potentially be helpful in the treatment of Insomnia, Addiction, Chronic Pain, and Multiple Sclerosis (MS). The scientific study supporting these alleged medicinal advantages of Cannabinoids is still in its initial stages.
Downloads
Article in PDF
Recent Articles
- New cancer drug tested in mice may benefit certain leukaemia patients
- Progenics got FDA Approval; Curcumin to cancer cells
- One of the leading causes of blindness, and a sight-threatening disease, Uveitis, now has novel t...
- The Business Cocktail
- Osivax raises USD 9M; Smyraf launched; Revolution secures USD 100M