Top 8 Breakthrough Gene Therapies for Retinitis Pigmentosa Treatment
Mar 31, 2026
Gene therapy is rapidly emerging as a transformative approach for retinitis pigmentosa treatment and other retinal degenerative disorders, with the eye providing an ideal environment for targeted interventions. Notably, the retina became the first tissue to receive a retinitis pigmentosa gene therapy FDA approval in the United States, marking a pivotal milestone in the retinal gene therapy market. To date, only one therapy, LUXTURNA retinitis pigmentosa, developed by Spark Therapeutics, has been approved, highlighting both progress and the unmet need for a broader retinitis pigmentosa cure.
Approved by the FDA in December 2017, LUXTURNA is a one-time gene therapy for retinitis pigmentosa designed for patients with biallelic RPE65 mutation-associated retinal dystrophy. It represents a major retinitis pigmentosa breakthrough, being the first approved gene therapy for a genetic condition, the first pharmacological option for inherited retinal diseases, and the first AAV-based therapy in the US. While Spark Therapeutics commercializes it in the US, Novartis leads its expansion internationally. This success has catalyzed innovation across the retinitis pigmentosa treatment market and broader retinal dystrophy treatment market.
According to Sadaf Javed, Manager of Forecasting at DelveInsight, LUXTURNA faces no direct competition in its approved indication. This exclusivity, Javed said, has allowed it to establish a strong foothold in the inherited retinal disease space, positioning it as the standard of care for eligible patients with RPE65-mediated vision loss. Its continued market leadership is further supported by robust clinical data and long-term follow-up demonstrating sustained efficacy and safety, Javed further added.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- BeiGene’s Brukinsa Approval; FDA Approval to Seagen’s TUKYSA; NICE Recommends Alnylam’s Amvuttra;...
- The promising Pipeline for Gene Therapy In Oncology
- Are you ready to embrace Gene Therapy?
- Is Gene Therapy the Next Cancer Treatment Revolution?
- AAV Vectors in Gene Therapy: How Recent Clinical Advances are Unraveling New Potentials?
Before this advancement, there were virtually no effective retinitis pigmentosa treatments, leaving patients with limited treatment for retinitis pigmentosa beyond supportive care. Today, gene and cell-based approaches are reshaping the landscape of RP treatment options, targeting key genes such as RHO, USH2A, and RPGR gene therapy pathways. Among these, USH2A remains a critical focus, with oligonucleotide-based therapies advancing in retinitis pigmentosa clinical trials. Additionally, optogenetics is gaining traction as a novel gene therapy retinitis pigmentosa approach, offering hope even in advanced stages where photoreceptor loss limits traditional strategies.
The pipeline for retinitis pigmentosa treatment options is robust, spanning early to late-stage clinical trials for retinitis pigmentosa. Prominent candidates include Botaretigene sparoparvovec (Johnson & Johnson Innovative Medicine/MeiraGTx), Laruparetigene zovaparvovec (Beacon Therapeutics), MCO-010 (Nanoscope Therapeutics), GS030 (Gensight Biologics), FT-002 (Frontera Therapeutics), HORA-PDE6b (EyeDNA Therapeutics), OCU400 (Ocugen), and BS01 (Bionic Sight), each contributing to the evolution of RP gene therapy and expanding possibilities for a future cure for retinitis pigmentosa. Alongside these innovations, research continues into medications for retinitis pigmentosa non-surgical, ensuring a diversified therapeutic approach.
Overall, the convergence of innovation in retinitis pigmentosa gene therapy clinical trials, optogenetics, and targeted molecular treatments signals a paradigm shift. As retinitis pigmentosa latest research advances, the field is moving closer to defining the best treatment for retinitis pigmentosa, with the ultimate goal of delivering durable, accessible, and potentially curative solutions.
Here, take a closer look at these gene therapies for retinitis pigmentosa treatment in detail.
Johnson & Johnson Innovative Medicine/MeiraGTx’s Botaretigene sparoparvovec
AAV5 gene therapy
Botaretigene sparoparvovec (bota-vec) is emerging as a high-potential retinitis pigmentosa gene therapy, specifically engineered to target the most common form of X-linked retinitis pigmentosa (XLRP) driven by mutations in the RPGR ORF15 gene. As momentum builds around gene therapy for retinitis pigmentosa, bota-vec is positioned at the forefront of innovation in the evolving retinal gene therapy market.
In a major strategic move, MeiraGTx partnered with Johnson & Johnson Innovative Medicine through an asset purchase agreement, covering full rights to bota-vec, alongside commercial supply and technology transfer agreements, signaling strong confidence in its role within the expanding retinitis pigmentosa treatment market and broader retinal dystrophy treatment market.
The therapy has already gained multiple regulatory boosts, including Fast Track and Orphan Drug Designation (ODD) from the FDA, along with PRIME, ATMP, and Orphan designations from the EMA, highlighting its promise as a potential breakthrough in X-linked retinitis pigmentosa treatment options and a step closer toward a cure for retinitis pigmentosa.
However, in May 2025, results from the Phase III LUMEOS study revealed that bota-vec did not meet its primary endpoint. Despite this, encouraging signals emerged, with subsets of patients demonstrating meaningful improvements in vision-guided mobility (VMA), keeping hope alive for this RPGR gene therapy in ongoing retinitis pigmentosa clinical trials.
Earlier, in March 2025, MeiraGTx had indicated that pivotal data from the LUMEOS trial, one of the most closely watched clinical trials for retinitis pigmentosa, would shape the future landscape of gene therapy retinitis pigmentosa and redefine treatment for retinitis pigmentosa.
As retinitis pigmentosa latest research continues to evolve, therapies like bota-vec underscore both the challenges and promise in the quest for the best treatment for retinitis pigmentosa, especially in comparison to existing medications for retinitis pigmentosa (non-surgical). While a definitive retinitis pigmentosa cure remains elusive, advancements in RP gene therapy are rapidly transforming the pipeline and redefining RP treatment options.
Beacon Therapeutics’ Laruparetigene zovaparvovec
AAV2 gene therapy
Laru-zova is emerging as a potential best-in-class contender in the race for a breakthrough retinitis pigmentosa gene therapy, positioning itself at the forefront of innovation in the evolving retinitis pigmentosa treatment market. Designed as a next-generation RPGR gene therapy, it is being actively developed as a transformative gene therapy for retinitis pigmentosa, a space long seeking a true retinitis pigmentosa cure.
Backed by multiple elite regulatory recognitions, including RMAT and Fast Track Designation (FTD) from the FDA, PRIME from the EMA, and ILAP from the UK MHRA, along with Orphan Drug Designation (ODD) in both the US and Europe, laru-zova stands out among retinitis pigmentosa treatments and signals strong momentum toward potential retinitis pigmentosa gene therapy FDA approval.
Beacon Therapeutics is strategically leveraging a robust clinical dataset spanning the Phase II/III VISTA trial, along with long-term evidence from the Phase II DAWN, Phase I/II HORIZON, and Phase II SKYLINE studies, to accelerate regulatory submissions across the US and Europe. This integrated approach reflects the growing maturity of the retinal gene therapy market and underscores the promise of gene therapy retinitis pigmentosa as a viable treatment for retinitis pigmentosa.
Adding to this momentum, in December 2025, Beacon announced dosing of the first patient in the LANDSCAPE trial, an open-label study evaluating bilateral administration, marking a critical step forward in retinitis pigmentosa clinical trials and expanding rp treatment options. Earlier, in July 2025, the company successfully completed enrollment in its pivotal VISTA trial, a landmark in clinical trials for retinitis pigmentosa. Topline 12-month data are anticipated in the second half of 2026, potentially shaping the future of retinitis pigmentosa latest research and redefining best treatment for retinitis pigmentosa.
As innovation accelerates across the retinal dystrophy treatment market, laru-zova represents a major retinitis pigmentosa breakthrough, offering hope beyond traditional medications for retinitis pigmentosa non-surgical and moving closer to a functional cure for retinitis pigmentosa. With multiple retinitis pigmentosa treatment options under investigation, this therapy could redefine standards in pigmentosa treatment and reshape the landscape of RP gene therapy worldwide.
Nanoscope’s MCO-010
AAV2-delivered optogenetic gene therapy
Nanoscope Therapeutics is turning heads in the retinitis pigmentosa treatment space with its game-changing candidate, MCO-010, a next-gen retinitis pigmentosa gene therapy designed to restore light sensitivity where it matters most. Delivered through a simple, well-established intraocular injection, this innovative gene therapy for retinitis pigmentosa introduces the ambient light-responsive MCO protein directly into retinal cells, offering new hope in the search for a retinitis pigmentosa cure.
With strong regulatory momentum, MCO-010 has secured both Fast Track Designation (FTD) and Orphan Drug Designation (ODD) from the U.S. FDA, putting it on a promising path toward retinitis pigmentosa gene therapy FDA approval. A rolling BLA is already underway, signaling a major leap forward in the evolving retinal gene therapy market and broader retinitis pigmentosa treatment market.
Globally, the therapy is gaining traction. The European Medicines Agency (EMA) has granted five Orphan designations, reinforcing its potential across multiple retinal dystrophy treatment market segments. Meanwhile, Japan’s Ministry of Health, Labour and Welfare (MHLW) has fast-tracked MCO-010 with both Sakigake and Orphan Drug status, highlighting its promise as a retinitis pigmentosa breakthrough and a leading RP gene therapy contender.
Backing the buzz is strong clinical evidence. In October 2025, positive three-year data from the REMAIN study, an extension of the Phase IIb/III RESTORE trial, demonstrated sustained benefits in patients, placing MCO-010 firmly among the most exciting retinitis pigmentosa clinical trials and gene therapy retinitis pigmentosa clinical trials underway today.
As the race intensifies for the best treatment for retinitis pigmentosa, MCO-010 stands out among emerging retinitis pigmentosa treatment options, including RPGR gene therapy and other medications for retinitis pigmentosa non-surgical approaches. With continued progress, it could redefine the future of treatment for retinitis pigmentosa, reshape retinitis pigmentosa latest research, and accelerate innovation across the retinal gene therapy market.
In a landscape hungry for effective retina pigmentosa treatment and scalable RP treatment options, MCO-010 is not just another candidate; it’s a potential paradigm shift toward a true cure for retinitis pigmentosa.
Frontera Therapeutics’ FT-002
rAAV gene therapy
FT-002 is emerging as a retinitis pigmentosa breakthrough, positioning itself at the forefront of gene therapy for retinitis pigmentosa. Designed specifically as an advanced RPGR gene therapy, it represents a next-gen approach in the evolving retinitis pigmentosa treatment market and broader retinal gene therapy market.
In a major regulatory milestone, FT-002 secured Orphan Drug Designation (ODD) from the FDA in January 2024, highlighting its potential as a future retinitis pigmentosa cure and one of the most promising retinitis pigmentosa treatments in development.
Momentum continued in September 2024, when Frontera announced that FT-002 received FDA clearance to advance into Phase II clinical trials in the US, marking a significant step forward in retinitis pigmentosa gene therapy clinical trials. This progress was backed by encouraging clinical data from earlier studies conducted in China, reinforcing its promise as a leading RP gene therapy candidate.
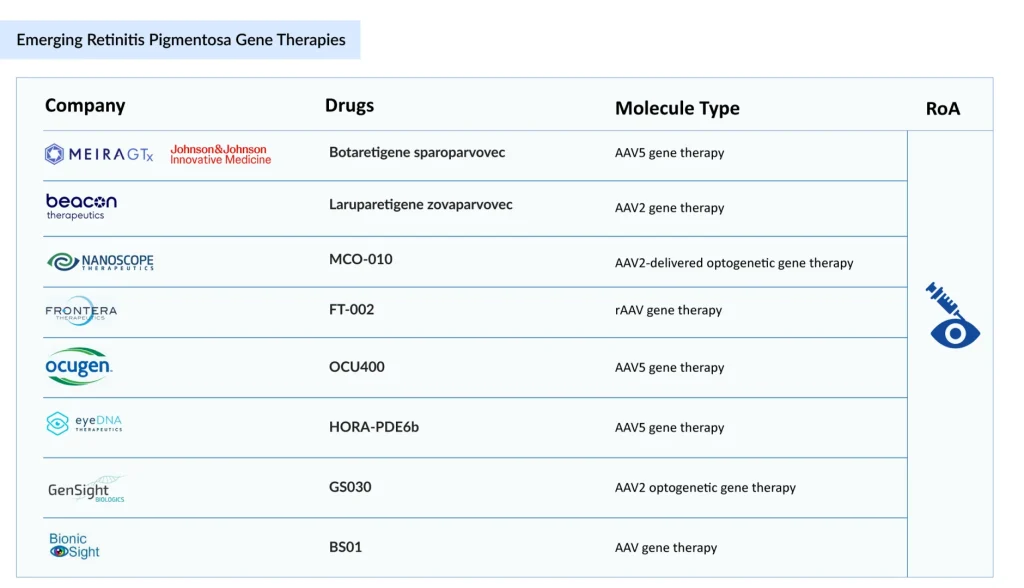
Gensight’s GS030
AAV2 optogenetic gene therapy
GS030 is redefining the future of retinitis pigmentosa treatment with a bold, next-gen approach that blends cutting-edge science and smart technology. This breakthrough retinitis pigmentosa gene therapy combines a bioengineered AAV2 7m8 vector with biomimetic goggles, working together to bring light back to vision.
At its core, GS030 leverages gene therapy for retinitis pigmentosa through optogenetics, delivering a gene that encodes a light-sensitive channelrhodopsin protein directly into retinal ganglion cells (RGCs). These cells, typically unaffected in retinal degeneration, are transformed into light-responsive units, offering a mutation-agnostic solution, something long sought in the retinitis pigmentosa treatment market and broader retinal gene therapy market.
Currently in retinitis pigmentosa clinical trials (Phase I/II for non-syndromic RP), GS030 represents a major retinitis pigmentosa breakthrough and a strong contender among emerging RP gene therapy and gene therapy retinitis pigmentosa innovations. As clinical trials for retinitis pigmentosa continue to evolve, GS030 stands out in the landscape of retinitis pigmentosa treatment options, especially among medications for retinitis pigmentosa non-surgical alternatives.
With growing momentum in retinitis pigmentosa latest research, GS030 is not just another entry; it’s a paradigm shift in treatment for retinitis pigmentosa, contributing to the expanding retinal dystrophy treatment market and reshaping expectations around retinitis pigmentosa treatments and RP treatment options.
EyeDNA Therapeutics’ HORA-PDE6b
AAV5 gene therapy
HORA-PDE6β is redefining the future of retinitis pigmentosa treatment, a next-gen AAV5-based innovation built to go straight to the root cause. This cutting-edge gene therapy for retinitis pigmentosa delivers a healthy, full-length PDE6β gene directly into the subretinal space, triggering rapid and robust protein expression in both rods and cones. The result? A powerful approach that could slow, or even stop, degeneration in patients, positioning it as a potential retinitis pigmentosa cure in the making.
As momentum builds in the retinal gene therapy market and retinitis pigmentosa treatment market, HORA-PDE6β stands out among emerging retinitis pigmentosa treatments and RP gene therapy innovations. Currently advancing through retinitis pigmentosa clinical trials (Phase I/II), it reflects the wave of retinitis pigmentosa breakthrough research shaping the future of care.
With growing interest in non-surgical medications for retinitis pigmentosa and evolving RP treatment options, therapies like HORA-PDE6β are at the forefront of retinitis pigmentosa latest research. While the journey toward retinitis pigmentosa gene therapy FDA approval continues, this program represents a promising step forward in gene therapy retinitis pigmentosa development.
Ocugen’s OCU400
AAV5 gene therapy
OCU400 is redefining the future of retinitis pigmentosa treatment with a bold, gene-agnostic approach. Developed by Ocugen, this cutting-edge retinitis pigmentosa gene therapy is powered by the nuclear hormone receptor gene NR2E3, a master regulator of retinal health. Instead of targeting a single mutation like traditional approaches (e.g., RPGR gene therapy), OCU400 works upstream to reset the entire dysfunctional gene network in RP patients, restoring balance across photoreceptor development, metabolism, phototransduction, inflammation, and cell survival.
This breakthrough positions OCU400 at the forefront of the evolving gene therapy for retinitis pigmentosa landscape, offering hope for a functional retinitis pigmentosa cure where few retinitis pigmentosa treatment options exist today. As part of the expanding retinal gene therapy market and retinal dystrophy treatment market, OCU400 is a strong contender for the best treatment for retinitis pigmentosa, especially among emerging medications for retinitis pigmentosa non-surgical.
In a major milestone for retinitis pigmentosa clinical trials, Ocugen announced in March 2026 that enrollment is complete for its pivotal Phase 3 liMeliGhT study. This one-year study marks a critical step toward retinitis pigmentosa gene therapy FDA approval, with topline data expected in Q1 2027. Positive results could accelerate BLA submission and bring this retinitis pigmentosa breakthrough to patients as early as 2027.
Importantly, the retinitis pigmentosa gene therapy clinical trials program has also gained global momentum, the European Medicines Agency (EMA) has accepted the U.S. trial data framework for future MAA submission, expanding OCU400’s reach in the global retinitis pigmentosa treatment market.
Bionic Sight’ BS01
AAV gene therapy
BS01 is redefining the future of retinitis pigmentosa treatment with a bold, next-gen approach to vision restoration. Developed at the Belfer Gene Therapy Core Facility at Weill Cornell Medicine, this cutting-edge retinitis pigmentosa gene therapy is built on GMP-grade innovation, bridging advanced science and clinical precision.
Early data from ongoing retinitis pigmentosa clinical trials are promising; patients have demonstrated the ability to perceive light and motion, with some even detecting direction. These findings place BS01 at the forefront of retinitis pigmentosa latest research and highlight its potential to reshape the retinitis pigmentosa treatment market and broader retinal dystrophy treatment market.
Currently in Phase I/II clinical trials for retinitis pigmentosa, BS01 is advancing the conversation around the best treatment for retinitis pigmentosa, especially for patients with limited non-surgical medications for retinitis pigmentosa.
As the field moves closer to retinitis pigmentosa gene therapy FDA approval, BS01 stands out as a pioneering force in gene therapy retinitis pigmentosa, offering new hope in the quest for an effective retina pigmentosa treatment and long-term vision restoration.
Beyond the frontrunners, the retinitis pigmentosa treatment pipeline is buzzing with innovation. Leading players like Neurotech Pharmaceuticals (NT-501), SparingVision (SPVN06, SPVN20, SPVN30), ViGeneron (VG901), PYC Therapeutics (VP-001), and Ray Therapeutics (RTx-015) are advancing cutting-edge candidates across multiple stages of retinitis pigmentosa clinical trials. From RPGR gene therapy to next-gen gene therapy for retinitis pigmentosa, the race toward a viable retinitis pigmentosa cure is accelerating.
The momentum in retinitis pigmentosa gene therapy is particularly striking. With encouraging safety profiles and early efficacy signals, these therapies are redefining what could become the best treatment for retinitis pigmentosa. As the industry edges closer to potential retinitis pigmentosa gene therapy FDA approval, the broader retinal gene therapy market and retinitis pigmentosa treatment market are poised for transformative growth. Alongside gene-based approaches, innovations in medications for retinitis pigmentosa non-surgical and stem cell strategies are expanding retinitis pigmentosa treatment options like never before.
Still, the journey toward a definitive cure for retinitis pigmentosa isn’t without hurdles. Challenges such as optimizing gene delivery systems, scaling gene therapy retinitis pigmentosa platforms, and addressing the lack of robust animal models continue to shape ongoing clinical trials for retinitis pigmentosa. Long-term efficacy, durability, and safety remain critical checkpoints for every emerging RP gene therapy and retinal dystrophy treatment market entrant.
Yet, the outlook is undeniably optimistic. With rapid strides in retinitis pigmentosa latest research and a surge in retinitis pigmentosa gene therapy clinical trials, the field is on the brink of a true retinitis pigmentosa breakthrough. As innovation converges across modalities, we are stepping into a new era, where treatment for retinitis pigmentosa is not just about slowing progression, but potentially restoring vision and redefining outcomes for patients worldwide.

Frequently Asked Questions
Prominent candidates include Botaretigene sparoparvovec (Johnson & Johnson Innovative Medicine/MeiraGTx), Laruparetigene zovaparvovec (Beacon Therapeutics), MCO-010 (Nanoscope Therapeutics), GS030 (Gensight Biologics), FT-002 (Frontera Therapeutics), HORA-PDE6b (EyeDNA Therapeutics), OCU400 (Ocugen), and BS01 (Bionic Sight), each contributing to the evolution of RP gene therapy and expanding possibilities for a future cure for retinitis pigmentosa.
Retinal gene therapies work by delivering a healthy copy of a defective gene directly into retinal cells, usually via viral vectors, so they can produce functional proteins again. This helps restore or preserve photoreceptor function, slowing degeneration and, in some cases, improving vision. By targeting the root genetic cause, these therapies aim to halt or even partially reverse vision loss in retinitis pigmentosa.
Recent clinical trial data for gene therapy in retinitis pigmentosa show encouraging but still early-stage outcomes. Optogenetic and gene-agnostic therapies (e.g., MCO-010) have demonstrated improvements in visual acuity, mobility, and light perception over ~1 year in small patient cohorts, with favorable safety profiles.
The only FDA-approved one is LUXTURNA (voretigene neparvovec-rzyl) for patients with biallelic RPE65 mutations. It’s a one-time gene therapy designed to restore vision in that specific genetic subtype.
Gene therapy for retinitis pigmentosa can carry risks such as immune reactions to the viral vectors used to deliver the gene, which may cause inflammation or reduce treatment effectiveness. There is also a possibility of unintended genetic effects or damage to retinal cells, potentially leading to vision complications. Additionally, long-term safety and durability of the treatment are still being studied.
Downloads
Article in PDF
Recent Articles
- Vertex & CRISPR Therapeutics Make History: FDA Approves exa-cel, the First CRISPR Gene Therap...
- FDA Approves LUMAKRAS with VECTIBIX for KRAS G12C-Mutated Colorectal Cancer; PYC Receives FDA Rar...
- Gene Therapy in Rare Disorders: Acceptance in Europe Faces Challenges
- The Future of Parkinson’s Disease Treatment: Unlocking the Potential of Cell and Gene Therapy
- BeiGene’s Brukinsa Approval; FDA Approval to Seagen’s TUKYSA; NICE Recommends Alnylam’s Amvuttra;...