How are Neurostimulation Devices Playing a Vital Role in the Treatment of Chronic Pain?
Jul 27, 2022
Table of Contents
Around 50 million people in the United States live with—and suffer from—chronic pain. The prevalence of chronic pain has grown significantly over the past few decades due to lifestyle changes, traumatic injury, a rise in critical surgeries, and growth in the overweight or obese population, among serval other underlying health conditions.
As per DelveInsight “Chronic Pain Epidemiology Forecast” report, the total prevalent case of chronic pain was more than 187 million in the 7MM in 2020, and it is expected to increase by 2032.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- FDA Approval to DePuy TELIGEN System; FDA Breakthrough Device Designation for the EndoStim System...
- ONWARD Medical’s ARC-EX System Gains CE Mark; Microbot Medical Secures FDA 510(k) Clearance for L...
- MIMEDX Launches HELIOGENTM Fibrillar Collagen Matrix; DocGo Launches Mobile X-ray Program; Labcro...
- How Metaverse is Set to Transform the Healthcare Dynamics?
- Vertex’s JOURNAVX: A Historic Milestone in Pain Treatment After a Long Dry Spell
Pain is classified as either acute or chronic. Acute pain is usually severe and short-lived, while chronic pain can range from mild to severe and present for extended periods (>3 months). If chronic pain persists for a long time, the patient may require treatment. While some pain patients receive relief from physical therapy, medication, or surgery, others aren’t helped by these treatments.
The next line of treatment often involves implantable neurostimulation devices that deliver electrical pulses (low-voltage electrical stimulation) to the spinal cord or peripheral nerves, interrupting pain signals before they reach the brain. While doctors have used neurostimulation for pain since the 1960s, neurostimulation devices and protocols continue to evolve in the quest to offer robust pain relief without side effects.
Neurostimulation delivers pain relief impulses by disrupting the pain signals traveling between the critical body parts, i.e., the spinal cord and the brain. Neurostimulation consists of components such as a Neurostimulator (which generates the electrical impulses), leads (insulated medical wires that deliver electrical pulses), and a programmer (to customize the neurostimulation system and its settings). Neurostimulation can be accomplished by both fully implantable and external radio-frequency powered systems. The patient or the user can alter the strength and location of stimulation with a handheld programmer, which is one of the major advantages of these devices. These Neurostimulation devices have several benefits as they can target the precise area where the patient or the user feels pain. These devices can be easily adjustable and are cost-effective as well.
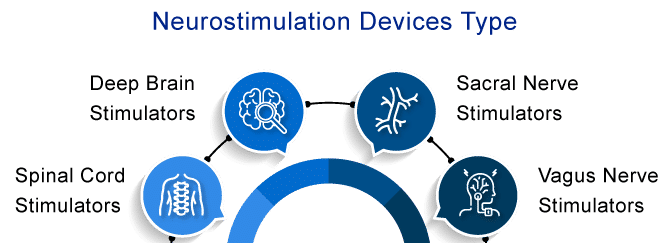
Types of Neurostimulation Devices
Neurostimulation may help many people manage pain more effectively. Doctors have prescribed several neurostimulation devices for over 40 years to control chronic pain and enhance the quality of life. The different types of neurostimulation devices used for chronic pain management include
Spinal Cord Stimulators
A spinal cord stimulator is an implanted device that emits low doses of electricity directly into the spinal cord to treat pain. Spinal cord stimulators are made up of thin wires (electrodes) and a compact, pacemaker-like battery pack (the generator). The electrodes are implanted between the spinal cord and the vertebrae (the epidural space), and the generator is placed beneath the skin, generally towards the buttocks or abdomen. When patients experience pain, spinal cord stimulators allow them to send electrical impulses using a remote control. Both the remote control and its antenna are located outside the body.
Spinal cord stimulation is most commonly employed when nonsurgical pain treatment methods have failed to offer enough relief. Spinal cord stimulators can be used to treat or manage various chronic pain conditions, including back pain, post-surgical pain, heart pain, complex regional pain syndrome, and amputation pain. As a result, the global spinal cord stimulators market has grown dramatically due to the growing burden of chronic diseases. Increased injuries and traumas following spinal procedures and changes in social behavior contribute to the growing demand for spinal cord stimulators.
As per DelveInsight analysis, the global spinal cord stimulators market is expected to grow at a CAGR of 8.70% to reach USD 3.72 billion by 2027.
The increased number of people suffering from chronic pain and neuropathic pain in the shoulder, legs, and knee is driving the demand for spinal cord stimulators. Moreover, technologically enhanced devices that are non-invasive eradicate specific aches, reduce patient suffering, and have no side effects are projected to drive growth in the global spinal cord stimulators market. Furthermore, favorable government laws for commercialization and product launch in the market, as well as an increasing elderly population, are some of the factors driving the growth of the spinal cord stimulators market.
Deep Brain Stimulators
Chronic intractable pain is incapacitating for people affected, affecting 5% of the population. Since the 1970s and 1980s, deep brain stimulation (DBS) has fallen out of favor as the centrally implantable neurostimulation of choice for chronic pain, with some neurosurgeons favoring motor cortex stimulation as the “last chance saloon.”
Deep brain stimulation offers an additional option for pain relief in some people. Central post-stroke pain, unusual facial pain, brachial plexus injury, and some patients who have failed spinal cord stimulators (SCS) are also signs. Except for some cases of facial pain, which may respond to peripheral nerve stimulation or high cervical stimulation, such conditions do not often react to SCS or DRG stimulation. There are three primary DBS target sites: (1) the thalamus- ventral posterolateral nucleus and ventral posteromedial nucleus (VPL/VPM); (2) the grey matter- Periventricular grey and periaqueductal grey (PVG/PAG); and (3) the newer target of the rostral anterior cingulate cortex (ACC) posterior to the anterior horns of the lateral ventricles. In those who have failed occipital nerve stimulation, a fourth target, the posterior hypothalamus, may be examined specifically for cluster headache.
As per our estimates, the global Deep Brain Stimulation Devices (DBS) Market was valued at USD 1.31 billion in 2021 and is expected to increase at a CAGR of 10.40% to reach USD 2.37 billion by 2027.
The growing prevalence of movement disorders such as Parkinson’s disease, the expansion of disease indications for DBS therapy, the rising geriatric population, and increased research and development activities in product development are expected to aid in the growing demand for DBS devices, leading to the growth of the deep brain stimulation devices market during the forecast period.
Vagus Nerve Stimulators
Vagus Nerve Stimulation (VNS) is an implanted stimulator, similar to a pacemaker-like device. Initially, it was designed to treat drug-resistant epilepsy, but today it has several new applications, including pain-relieving. Vagus Nerve Stimulators come with an implantable programmable generator embedded in the chest wall and delivers electrical impulses to the vagus nerve in the neck. The Vagus Nerve is one of the longest cranial nerves that ranges from the brainstem to the colon. The devices stimulate the vagus nerve and provide relief to patients.
Vagus nerve stimulators come in two types: conventional implanted devices & non-invasive unimplanted devices. Non-invasive Vagus Nerve Stimulators are found to have a more significant impact in treating acute migraines and cluster headaches. Several Vagus Nerve stimulations are approved by healthcare agencies worldwide. In the US, Vagus Nerve Stimulation is approved for treating epilepsy and treatment-resistant major depressive disorder. Similarly, some non-invasive devices are available to deal with episodic cluster headaches and migraine pain. As the prevalence of neurological disorders is increasing, the popularity and demand for non-invasive Vagus Nerve Stimulators devices are also growing.
As per DelveInsight, the Global Vagus Nerve Stimulation Market was valued at USD 426.22 million in 2021 and growing at a CAGR of 9.06% during the forecast period (2022 to 2027), it is expected to reach USD 717.17 million by 2027.
The rising prevalence of headache disorders such as migraine, and others, the increasing popularity of Vagus Nerve Stimulators therapy as an alternative or even conjunctive treatment in cases where drug resistance develops, and growing research in studying the effects of Vagus Nerve Stimulators on other indications such as tinnitus and chronic pain besides the approved ones, technological advancements leading to the development of new products such as non-invasive Vagus Nerve Stimulators devices thereby are expected to stimulate the Vagus Nerve Stimulation market growth in the coming years.
Sacral Nerve Stimulators
The sacral nerves consist of five pairs of nerves that exit the spinal column at its lowest point, the sacral vertebral area. To provide effective treatment options, Sacral Nerve Stimulators have emerged as a potential option to overcome the pain associated with the Sacral Nerve. Over the past few years, the application of sacral nerve stimulation as a treatment option has grown immensely for conditions such as urinary retention, urinary incontinence, anal incontinence, and constipation, among others. People with urgency-frequency sometimes may experience chronic pelvic pain.
Sacral nerve stimulation involves electrical stimulation of a sacral nerve root using an electrode and a stimulator. Sacral Nerve Stimulators are also found to be a safe and effective option for treating intractable pelvic pain with incomplete cauda equina syndrome (CES). Although the mechanism of action of neuromodulation remains unexplained, however, studies so far have shown that it is a treatment with efficacy and durability.
Key Companies in the Neurostimulation Devices Market
Companies in the industry obtained financing and investments from a few government bodies to conduct novel research in the field of neurostimulation devices. Product certifications from regulatory agencies such as the FDA, PMDA, Japan’s Ministry of Health, Labour, and Welfare, CE mark certification, European Medical Device Regulation (MDR) certification, and TGA clearances were granted to the companies.

Some of the key market players operating in the neurostimulation devices Market include Medtronic PLC, Cyberonics, Boston Scientific Corporation, Abbott Laboratories, Aleva neurotherapeutics SA, ElectroCore Inc., EndoStim Inc., Nevro Corporation, and others.
Neurostimulation Devices Market Dynamics
The major cause of disability is neurological disorders, including stroke, brain, and other nervous system malignancies, traumatic brain injury, spinal cord injury, Alzheimer’s disease, other dementias, Parkinson’s disease, epilepsy, migraine others. Strokes are the world’s second greatest cause of death. Over the last 25 years, the sheer number of people affected by, or still disabled by, neurological disorders have increased significantly, which in turn drives the neurostimulation devices market.
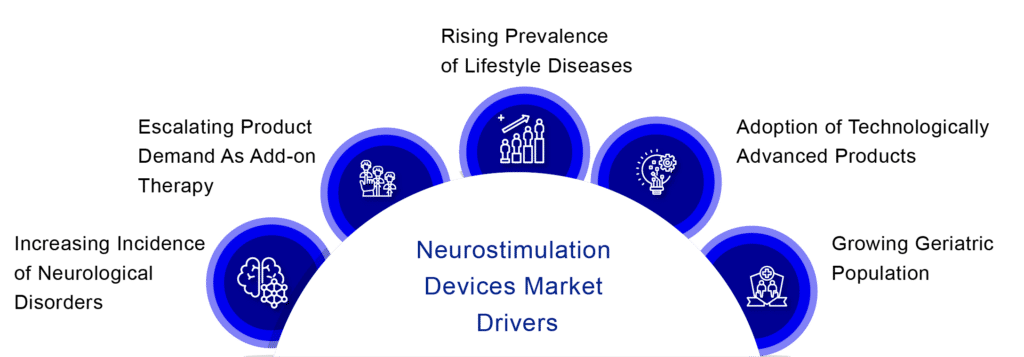
Furthermore, companies are increasing their research and development to develop more effective and simple products. The availability of restricted treatment options for the disease, combined with high product efficacy demonstrated in treating neurological diseases, is likely to drive neurostimulation devices’ market expansion throughout the forecasted period. Moreover, rising depression instances are also expected to contribute to neurostimulation devices market growth since neurostimulation devices allow simple treatment by activating or suppressing electrical brain signals.
However, certain factors, such as surgical problems following device insertion, high product costs, low awareness in low-income countries, high risks of infection, and product recall, represent a modest barrier to the expansion of the neurostimulation devices market.
As per DelveInsight estimates, the global Neurostimulation Devices market is anticipated to grow at a CAGR of 11.77% during the forecast period from 2022 to 2027, to reach USD 11.34 billion by 2027.
The increasing incidence of chronic diseases such as migraine and epilepsy, rising product demand as add-on therapy, the prevalence of lifestyle diseases such as depression and chronic pain, adoption of technologically advanced products, and the rising geriatric population are driving the demand for neurostimulation devices market.
Key Developments in the Neurostimulation Devices Market
Over the past few years, several key developments have been observed in the Neurostimulation Devices Market. The segment has registered major mergers & collaborations, breakthroughs, and investments. Some of the key development in the Neurostimulation Devices Market are as follows –
- In July 2021, Unity HA received Breakthrough Device Designation by FDA for the Pulsante® SPG Microstimulator System to treat acute pain associated with chronic cluster headaches.
- In July 2021, Nevro Corp. received FDA approval for its Senza® System for treating chronic pain associated with Painful Diabetic Neuropathy (PDN).
Similarly, other key developments have taken place in recent times. With the rising demand for Neurostimulation Devices, the growing awareness, and favorable approaches by the government to approve devices, the market is expected to observe phenomenal growth in the upcoming years.
Neurostimulation Treatment for Chronic Pain Management – Future Perspective
The prevalence of chronic pain has grown significantly worldwide over the past few decades due to lifestyle changes and a rise in injuries, among others. According to the American Academy of Pain Medicine, more than 1.5 billion people worldwide have chronic pain. The overall prevalence is expected to rise in the coming years.
Chronic Pain is associated with long-term disability and results in a significant decrease in productivity and health outcomes. If it remains untreated or ignored for a long time, it may lead to several physical and psychosocial effects on the body. It can often also result in several other diseases, including depression.
Neurostimulation has emerged as a potential treatment option for chronic pain management, which is cost-effective and easy to use. It has immense potential to replace the conservative treatment approaches available in the market today. As the technology evolves, Neurostimulation devices are expected to observe significant growth in the coming years.

Downloads
Article in PDF
Recent Articles
- Noninvasix’s LIVOx™ Central Venous Oxygenation Monitor; LivaNova’s aura6000 System; Oxford...
- C2Dx’s Shaw Scalpel System; Texas Original’s RSO Gummies; Abbott’s Whole Blood Rapid Test FDA Cle...
- Nexalin Technology Secures Israeli Regulatory Clearance for Gen-2 SYNC 15 mA DIFS™ Neurostimulati...
- Merck sells biosimilars; AbbVie’s PARP; Pharma heads; Biogen looks to M&A
- Alma Announced the Launch of Alma Duo; Phillips-Medisize Launched a Pen Injector Platform; FDA Ap...