Diabetic Retinopathy: The Hornet’s Nest of Diabetes Mellitus
Jun 03, 2022
Table of Contents
Diabetic Retinopathy (DR), a microvascular disease, is an eye condition in diabetic patients, affecting blood vessels in the retina and causing significant visual loss in working populations. The retina needs a constant supply of blood, which it receives through a network of tiny blood vessels. Constant high blood sugar levels damage these blood vessels, making it the most common and severe ocular complication. The propensity to develop diabetic retinopathy is directly proportional to the patient’s age and duration of diabetes, as well as poor glycemic control and fluctuating blood pressure levels. Poor glycemic control, uncontrolled hypertension, dyslipidemia, nephropathy, male sex, and obesity worsens diabetic retinopathy. Typical fundus features of diabetic retinopathy include microaneurysms, hard exudates, macular edema (diabetic macular edema or DME), and new vessels (in proliferative DR or PDR). Although there is no permanent cure found for the disease, anti VEGFs are proven to act as symptomatic Diabetic Retinopathy cure.
A serious sight-threatening complication of Diabetes – Diabetic Retinopathy symptoms includes spots or dark strings floating in your vision (floaters), blurred and fluctuating vision, dark or empty areas in your vision as well as vision loss. There are 4 Diabetic Retinopathy Stages including Mild Non-Proliferative Diabetic Retinopathy, Moderate Non-Proliferative Diabetic Retinopathy, Severe Non-Proliferative Diabetic Retinopathy, and Proliferative Diabetic Retinopathy.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Japan is transforming its Healthcare sector through Regenerative Medicine
- Cancer, diabetes and eczema on list; AbbVie’s Humira; Teva weighs $2B; Fresenius in buyout ...
- Orthofix Launched the Galaxy Fixation Gemini System; Oxford Biodynamics Launched the EpiSwitch Pr...
- Can Cannabinoids be an Effective Medicinal Substance?
- Understanding The Increasing Prevalence Of Diabetes And Its Complications
Diabetic Retinopathy risk factors include independent aspects such as hypertension, duration of diabetes, diabetic neuropathy, diabetic nephropathy, diabetic foot ulcer, foot amputation, fasting blood glucose, serum total cholesterol, serum triglyceride, and HbA1c. Retinal photography and digital imaging are considered to be an effective technique for Diabetic Retinopathy screening. Over time, Diabetes damages small blood vessels throughout the body, including the retina. Diabetic Retinopathy occurs when these tiny blood vessels leak blood and other fluids. Diabetic Retinopathy causes the retinal tissue to swell, resulting in cloudy or blurred vision. Diabetic Retinopathy usually affects both eyes. The longer a person has diabetes, the more likely they will develop Diabetic Retinopathy. If left untreated, Diabetic Retinopathy can even cause blindness.
Diabetic retinopathy is now recognized as an inflammatory neurovascular complication of the systemic disease with neuronal injury/dysfunction preceding the current clinical microvascular recognized damage. Furthermore, it is indicative of the inflammatory tissue injury concurrent in other organs.
Calibration of Diabetic Retinopathy Treatment
Diabetic Retinopathy severity ranges from Non-Proliferative Diabetic Retinopathy (NPDR) to Proliferative Diabetic Retinopathy (PDR); with NPDR, the initiation of blurry vision starts. Neovascularization leads to vision blockage on progression toward PDR (the more advanced stage of diabetic eye disease). With the development of PDR, many patients develop vision-threatening complications, including diabetic macular edema (DME) and neovascularization that can lead to blindness.
Therapeutic Interventions in Diabetic Retinopathy
Current therapeutic paradigms focus on treating an advanced disease once Proliferative Diabetic Retinopathy or Diabetic Macular Edema has shown its development in Diabetic Retinopathy patients.
At present, Diabetic Retinopathy treatment options include anti-VEGF medications and steroid agents, which slow down disease progression leading to improved vision. Three anti-VEGF drugs are preferred as the first line of therapy. They work by blocking the production of new blood vessels and reducing macular swelling. This slows vision loss and improves vision. Approved anti-VEGF therapies (first line of therapy), including aflibercept (Eylea from Regeneron and Bayer), ranibizumab (Lucentis from Genentech and Novartis), and bevacizumab (Avastin from Genentech); dominate the diabetic retinopathy treatment landscape. Anti-VEGF therapies are highly effective and viable in regressing retinal neovascularization in eyes with Proliferative Diabetic Retinopathy.
Race to be the First Mover
Diabetic Retinopathy diagnosis relies on the detection of microvascular lesions. The advent of anti-VEGF therapy has shown significant clinical benefits to patients with Diabetic Retinopathy. In clinical practice, anti-VEGF drugs are limited due to the need for frequent injections, financial burden, and poor patient compliance. Intravitreal corticosteroids have also shown clinical benefits in treating patients who do not respond to refractory DME or anti-VEGF therapy. Diabetic Retinopathy treatment remains a challenge till now. However, it remains difficult for patients to achieve reading or driving vision with currently available therapies.
Optimizing current therapies regarding the number, dose, and duration of intravitreal injections and combination therapy strategies is critical for improving the quality of life of Diabetic Retinopathy patients. Further investigation is needed to understand better Diabetic Retinopathy etiology. Greater efforts are needed to facilitate the transfer of new research from the bench to the bedside.
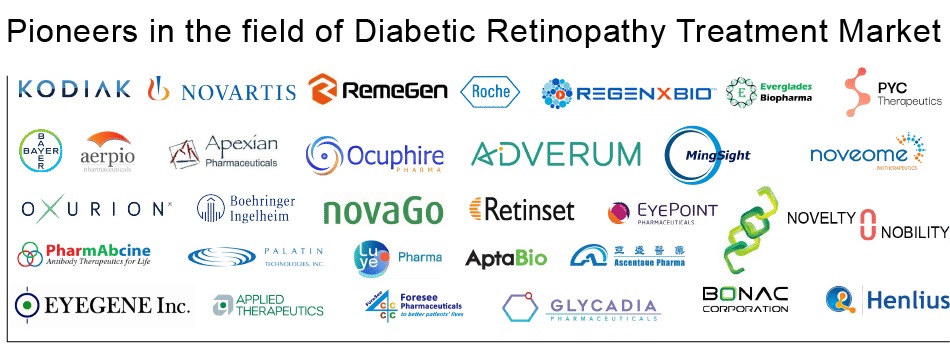
The current understanding of Diabetic Retinopathy has greatly improved in recent decades, leading to growing awareness, improved management, and better outcomes. Some of the key Diabetic Retinopathy Pipeline companies working to develop potential drug candidates to improve the Diabetic Retinopathy treatment scenario include names like Kodiak Sciences, Opthea Limited, Novartis, Bayer, Allergan, Graybug Vision, RemeGen, Aerpio Therapeutics, Hoffman-La-Roche, Regenxbio Inc., Apexian Pharmaceuticals, Inc., Ocuphire Pharma, Inc., Adverum Biotechnologies, Oxurion, MingSight Relin Pharmaceuticals, Boehringer Ingelheim, NovaGo Therapeutics, RIBOMIC, Retinset SL, PYC Therapeutics, PharmAbcine, Palatin Technologies, LUYE PHARMA, Curacle, BONAC corporation, AptaBio Therapeutics, Ascentage Pharma, EyeGene, Applied Therapeutics, Foresee Pharmaceuticals, Glycadia, Everglades Biopharma, Novelty Nobility Inc, Noveome Biotherapeutics, Shanghai Henlius Biotech, EyePoint Pharmaceuticals, and several others. Furthermore, some pharmaceutical companies are also trying to be part of the intervention by actively entering collaborations with other players.
Early Victories
Till date, there are 3 FDA approved drugs for Diabetic Retinopathy treatment. In May 2019, the US FDA approved Regeneron Pharmaceutical’s Eylea (aflibercept) injection for all stages of Diabetic Retinopathy treatment, thereby reducing the risk of blindness. Eylea is expected to lose its patent by 2025, which would lead to biosimilars launch into the market causing the shift in patient pool towards cost-effective options.
Bevacizumab, sold under the brand name Avastin, produced by Genentech is a medication used to treat several types of cancers and specific eye diseases. Bevacizumab is used as an off-label for Diabetic Retinopathy. It is on the World Health Organization’s List of Essential Medicines.
FDA approved ranibizumab, another drug produced by Genentech in 2017 for all stages of Diabetic Retinopathy treatment. Ranibizumab is sold under the trade name Lucentis. It is a monoclonal antibody fragment (Fab) created from the same parent mouse antibody as bevacizumab. Its effectiveness is similar to that of bevacizumab. Ranibizumab was developed by Genentech and marketed by them in the United States, and elsewhere by Novartis, under the brand name Lucentis. The total market size of Ranibizumab is expected to decrease by 2032 as the patent of Lucentis expired in 2020 in the US, and its biosimilars are already available in the market.
Diabetic Retinopathy Market and Epidemiology
According to secondary research by analysts at DelveInsight, it is estimated that Diabetic Retinopathy prevalence is slightly higher in males as compared to females. Diabetic Retinopathy epidemiology segmentation can be done on the basis of total prevalent cases of Diabetic Retinopathy, gender-specific cases of Diabetic Retinopathy, Severity-specific cases of Diabetic Retinopathy, and Age-specific cases of Diabetic Retinopathy.
Analysts at DelveInsight the combined Diabetic Retinopathy market size in the 7MM is anticipated to grow at a significant CAGR (Compound Annual Growth Rate) in the coming years. There are many innovative key Diabetic Retinopathy players involved in the development of promising therapeutic products such as Opthea Limited, Regenxbio, Kodiak Sciences Inc., Novartis, Graybug Vision, and others whose presence will be a key factor for Diabetic Retinopathy market growth in the coming years.
To Get Rich Insights on the Market of Diabetic Retinopathy, Visit our Infographics @ Diabetic Retinopathy Market
Emerging Therapies in Diabetic Retinopathy
At present, the Diabetic Retinopathy pipeline seems to be extremely robust. The key Diabetic Retinopathy therapies in several development phases are OPT-302, RGX 314, KSI-301, APX3330, OCS-01, and many others. Out of many, some of the Diabetic Retinopathy pipeline drugs in different phases of development under clinical trials include –
OPT-302 is currently under investigation by Opthea Limited. It is a first-in-class VEGF-C/D inhibitor. It is a soluble fusion protein comprising immunoglobulin-like domains 1 to 3 of the extracellular domain of vascular endothelial growth factor receptor 3 (VEGFR-3) fused to the Fc fragment of human immunoglobulin G1 (IgG1).
RGX-314 is being investigated by REGENXBIO Inc. as a potential one-time treatment for wet AMD, diabetic retinopathy, and other chronic retinal conditions. RGX-314 consists of the NAV AAV8 vector, which encodes an antibody fragment designed to inhibit vascular endothelial growth factor (VEGF). RGX-314 is believed to inhibit the VEGF pathway by which new, leaky blood vessels grow and contribute to the fluid accumulation in the retina.
KSI-301 is a novel anti-VEGF biologic built on a propriety antibody biopolymer conjugate (ABC) platform by Kodiak Sciences. KSI301 is given as an intravitreal injection and is expected to inhibit VEGF for up to 6 months continuously. The unique properties of KSI 301 are intended to provide patients with long-term control of DME and improve visual results while reducing the burden of frequent anti-VEGF injections. In addition, KSI301 is designed to prevent and reverse the progression of DR with long-term efficacy and may reduce the risk of vision-threatening complications from DR.
Brolucizumab (RTH258) is a humanized single-chain antibody fragment (scFv) produced by Novartis. It is considered to be the most clinically advanced, humanized single-chain antibody fragment to reach this stage of development. Single-chain antibody fragments are highly sought after in drug development due to their small size, enhanced tissue penetration, rapid clearance from systemic circulation, and drug delivery characteristics.
Expected Roadblocks and Competition
Eylea (aflibercept) has seen a raft of new competitors encroach on its territory till very recently. Eylea recorded its best performance in terms of volume and sales, and it is carving out a new niche of its own by further penetrating the diabetic eye disease landscape.
Diabetic Retinopathy is a disease of the neurovascular unit, with multiple, interdependent cell types contributing to the dysfunction of the retina. New therapeutic approaches should adopt a more holistic view of how diabetes affects the retina and tailor appropriate treatments to more precisely defined disease phenotypes with the exciting prospect of achieving successful clinical outcomes for all patients.
Find Out Comprehensive Details on the Disease Through Our Newsletter – Diabetic Retinopathy Newsletter
Way Ahead
Just in the past few years the understanding of Diabetic Retinopathy has greatly improved leading to growing awareness, improved management, and better outcomes. Diabetic Retinopathy market size is anticipated to increase in the coming years owing to factors such as extensive research and development activities of pharmaceutical companies, along with the launch of various multiple-stage Diabetic Retinopathy pipeline products will definitely dominate in the near future.